Compressibility factor (Z) for a van der Waals real gas at critical point is
$ 5.00 · 4.6 (517) · In stock

Share your videos with friends, family and the world

Non-Ideal Gas Behavior Chemistry: Atoms First
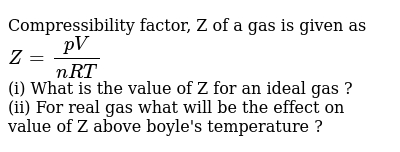
Compressibility factor, Z of a gas is given as Z=(pV)/(nRT) (i) What

plotting - How to plot Compressibility factor Z vs Pressure P using ParametricPlot? - Mathematica Stack Exchange

Compressibility factor - Wikipedia
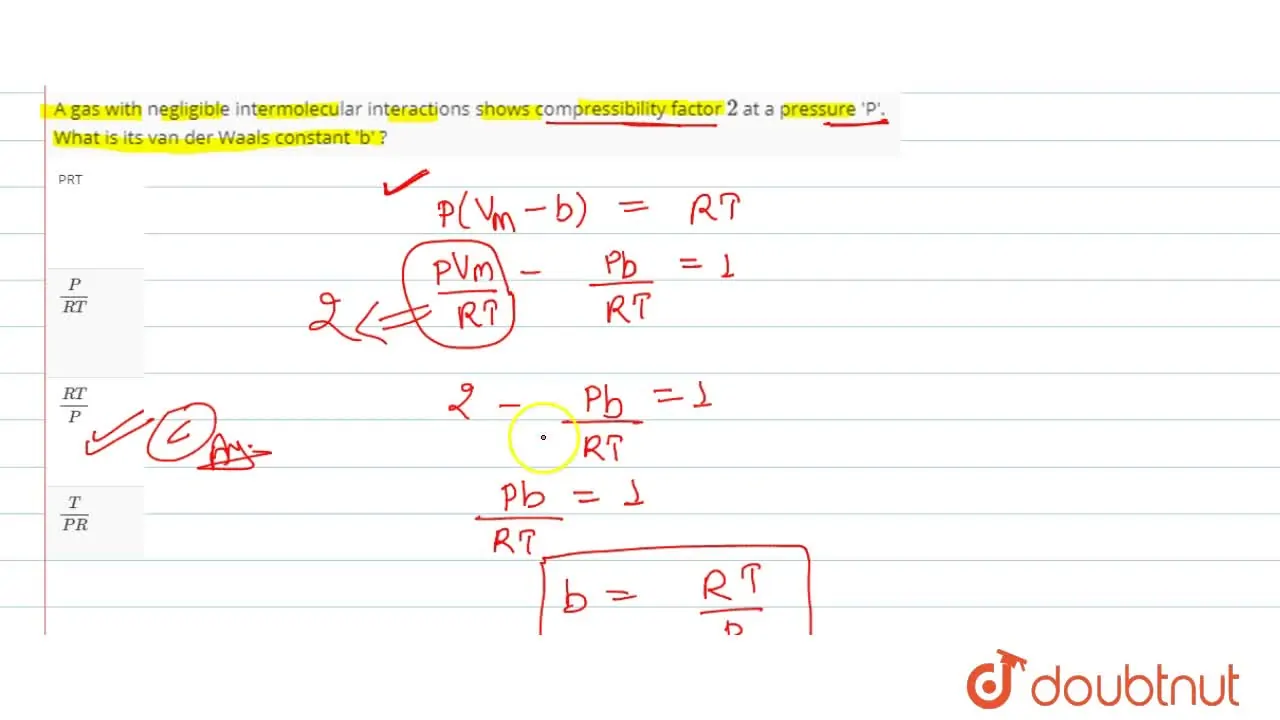
A gas with negligible intermolecular interactions shows compressibilit
Solved Find the compressibility factor Z for a real gas with

Compressibility factor, Z of a gas is given as Z=(pV)/(nRT) (i) What

Van der Waals Equation - Derivation, Relation Between Ideal Gas Law, Application

Van Der Waals Equation of State - an overview

Under critical conditions, the compressibility factor for a gas is .

The critical pressure P(C) and critical temperature T(C) for a gas obe

PhysChemistry for ChemEng 005] Real Gases Flashcards
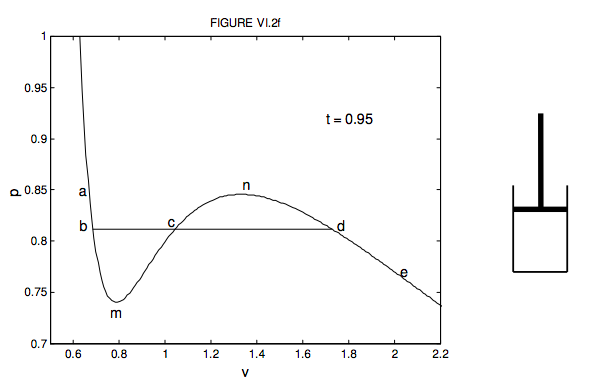
6.3: Van der Waals and Other Gases - Physics LibreTexts
