At a given temperature T gases Ne Ar Xe and Kr are found to deviate from ideal gas behavior (JEE MAINS 2019) - Doctor Logics Sunny Garg Chemistry
$ 13.99 · 4.6 (517) · In stock

At a given temperature T, gases Ne, Ar, Xe and Kr are found to deviate from ideal gas behavior. Their equation of state is given as P=RTV−b at T. Here, b is the van der Waals constant. Which gas will exhibit steepest increase in the plot of Z (compression factor) vs P?
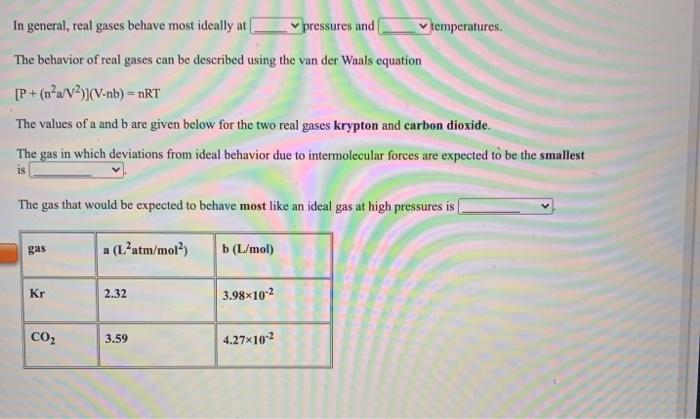
Solved In general, real gases behave most ideally at

Thermodynamics and Chemical Energitics- JEE Main MCQ'S & Solutions
Solved In an ideal gas thermometer, the relation PV=nRT
Sol Gel Book, PDF, Solid Oxide Fuel Cell
For a certain gas which deviates a little from ideal behaviour, the values of density, rho were measured different values of pressure, P. The plot of P/rho on the Y-axis versus P

The temperature of an ideal gas is increased from 27^(@)C to 127^(@)C

Identity of Thermodynamic Temperature Scale with the Perfect Gas

Solved 4.11 Nenideal Gias Feuatien! axi. where R is the

Solved In general, real gases behave most ideally at

At a given temperature T, gases Ne, Ar, Xe and Kr are found to deviate from ideal gas behaviour.

At a given temperature T, gases Ne, Ar, Xe and Kr are found to deviate

At a given temperature T, gases Ne, Ar, Xe and Kr are found to deviate from ideal gas behaviour. Their equation of state is given as P=RTV b at T.Here, b is