What is the change in internal energy (in J) of a system that
$ 11.00 · 5 (254) · In stock

I found an increase of 3100J Have a look
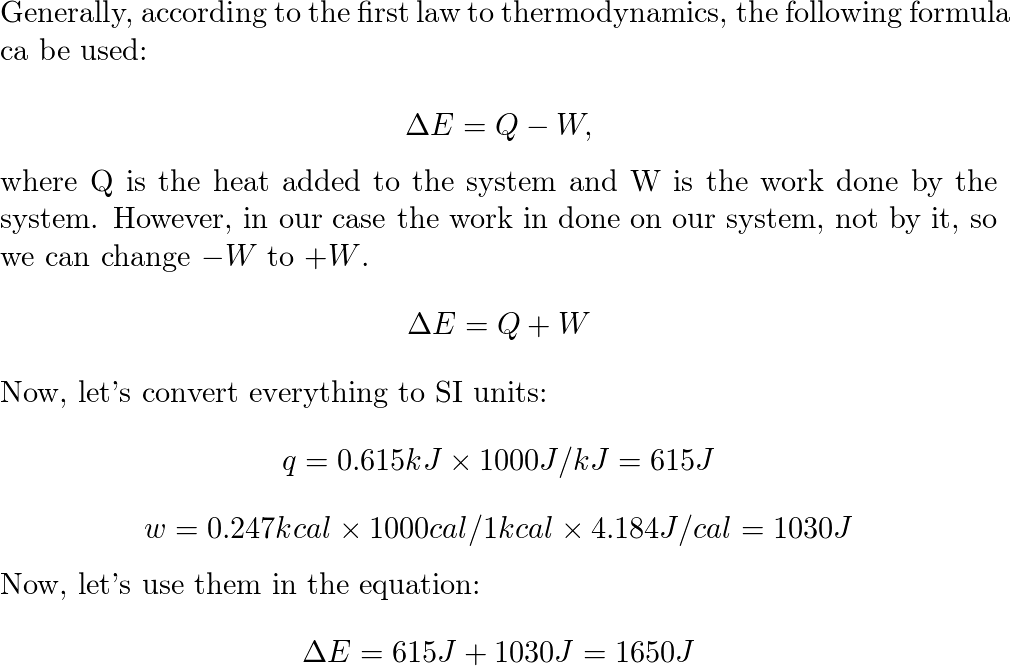
What is the change in internal energy (in J) of a system tha
53. During a process a system absorbs 710 J of heat and does work? The change in internal energy for the process is 460 J. What is the work done by the system?
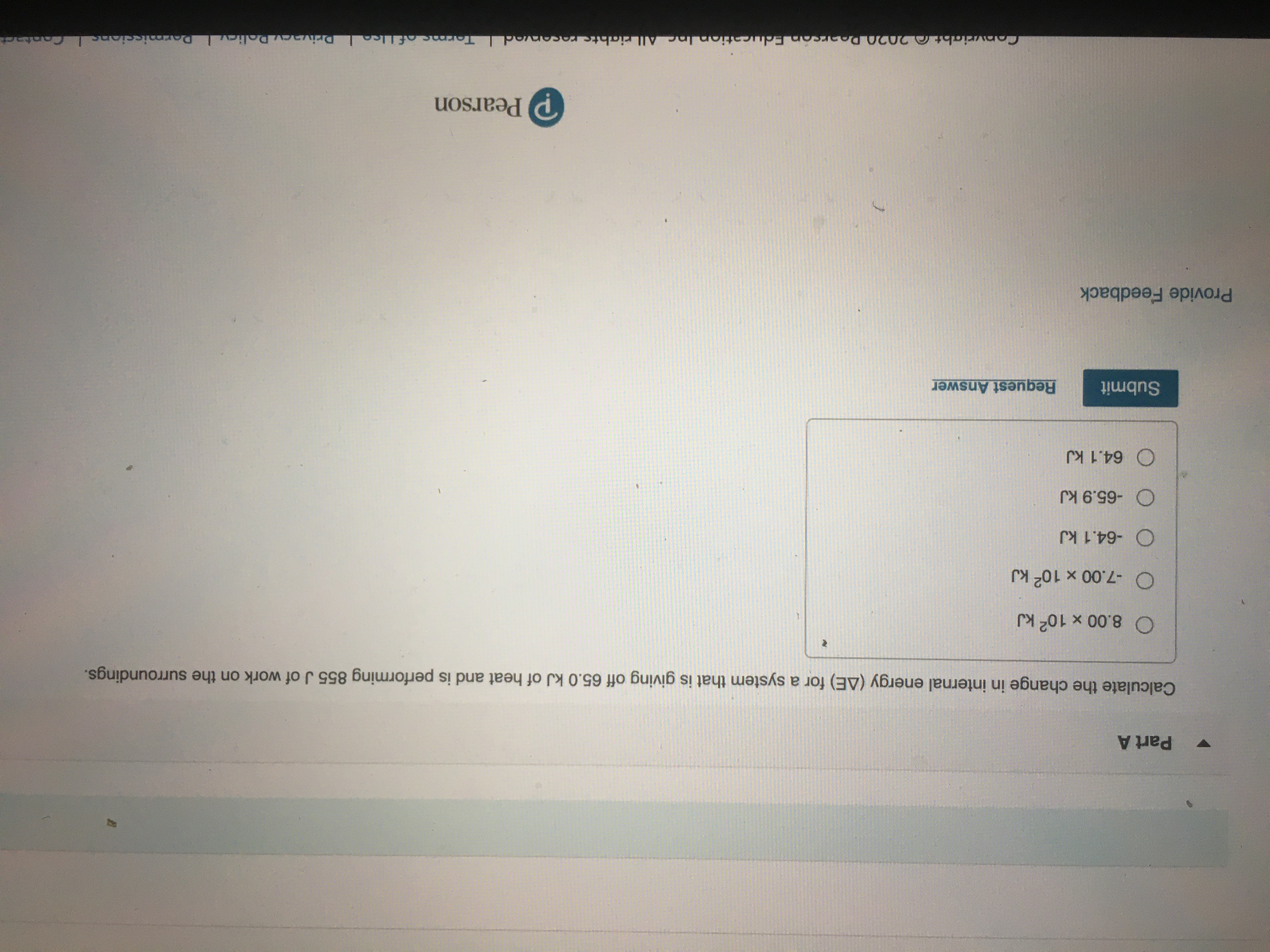
Answered: Calculate the change in internal energy…

Ch6.1 The Nature of Energy (hustle!) - ppt download

500 J of heat was supplied to a system constant volume. It resulted in the increase of temperature of the system from 20^oC to 25^oC. What is the change in internal energy

Tutorial 2- Thermodynamic - CHM431 TUTORIAL 2 : THERMODYNAMIC SEM

400 J of heat is added to a system. If the change in internal

Calculate the change in internal energy delta E for a system that is giving off 25 0kJ of heat and i

L 20 Thermodynamics [5] heat, work, and internal energy heat, work, and internal energy the 1 st law of thermodynamics the 1 st law of thermodynamics the. - ppt download
How to calculate ΔE when the system absorbs 250 J of heat energy

Internal Energy, Heat, and Work Thermodynamics, Pressure & Volume
Calculate the change in internal energy of a system if the energy of the surrounding get increased by 200j and a machine perform work of 500j on the system
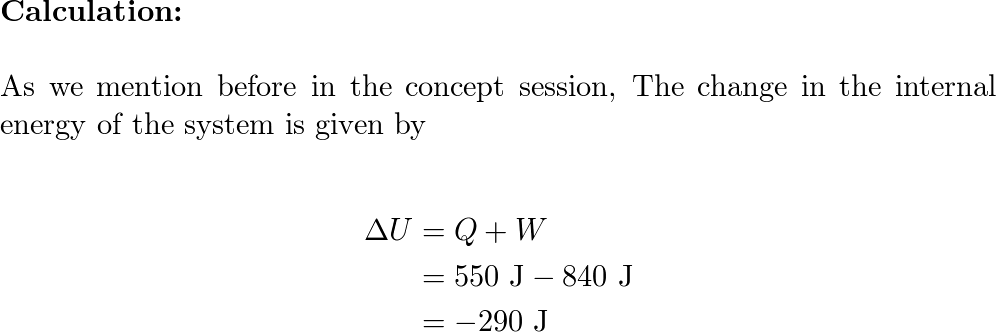
A system takes in 550 J of heat while performing 840 J of wo