Compression of a gas due to external pressure and the
$ 18.99 · 5 (182) · In stock

Solved A gas is compressed from an initial volume of 5.35 L

12.2 First law of Thermodynamics: Thermal Energy and Work

Waldo QUIROZ, Professor (Full), PhD Chemistry

PDF) Natural laws and ontological reflections: the textual and

Cristian MERINO RUBILAR, Professor (Assistant)

Cristian MERINO RUBILAR, Professor (Assistant)
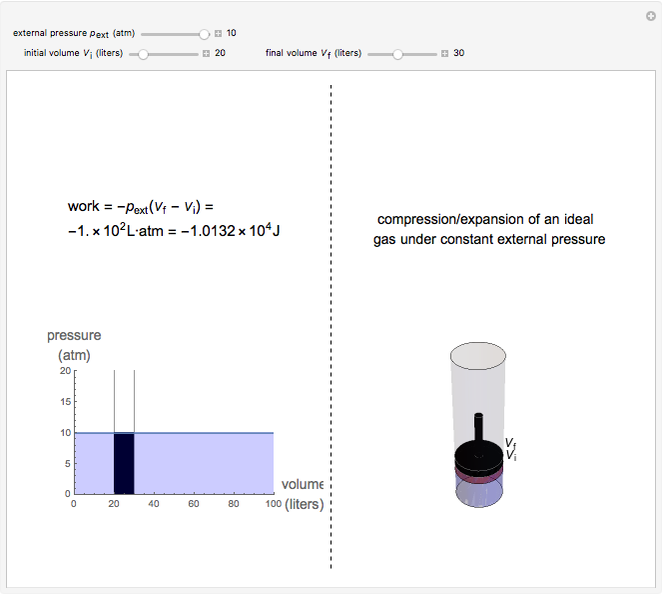
Isobaric Compression and Expansion of an Ideal Gas - Wolfram Demonstrations Project
Natural laws and ontological reflections: the textual and didactic

SOLVED: A gas compressed from a volume 0f 9.45 L to 8.23 L under constant external pressure 0f 5.60 atm How much work done on the gas joules? Give the value t0

What will be the work done on an ideal gas enclosed in a cylinder, when it is compressed by a constant external pressure, pext in a single step as shown in Figure
thermodynamics - Are you supposed to use the internal or external pressure for the $pV$ work integral? - Physics Stack Exchange

How will you calculate work done on an ideal gas in a compression, when change in pressure is ca
Solved A gas is compressed from an initial volume of 5.40 L