If `Z` is a compressibility factor, van der Waals' equation at low
$ 23.00 · 4.8 (73) · In stock

If `Z` is a compressibility factor, van der Waals' equation at low pressure can be written as

Compressibility Factor of Gas Overview, Equation & Chart

Under critical conditions, the compressibility factor for a gas is .
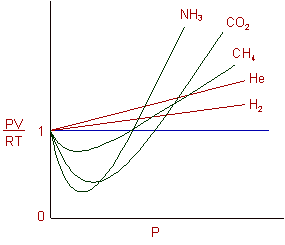
REAL GASES, DEVIATION FROM IDEAL GAS BEHAVIOUR
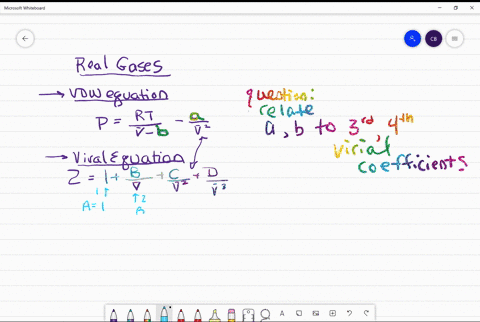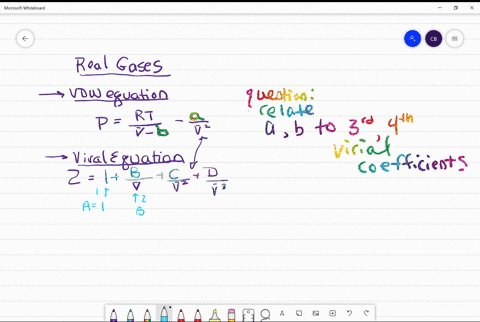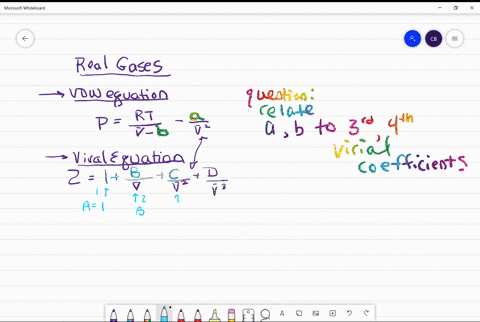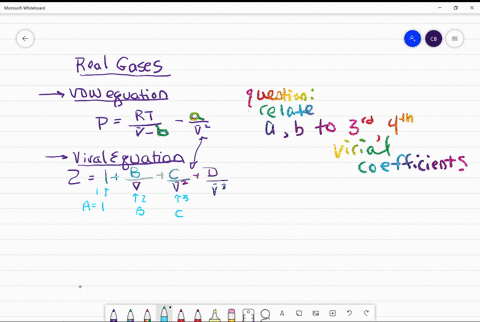
⏩SOLVED:Use the van der Waals constants for CH4 in Table 1.3 to

What is the compressibility factor (Z) for 0.02 mole of a van der

image.slidesharecdn.com/unit10realgasesvdwfl14fina
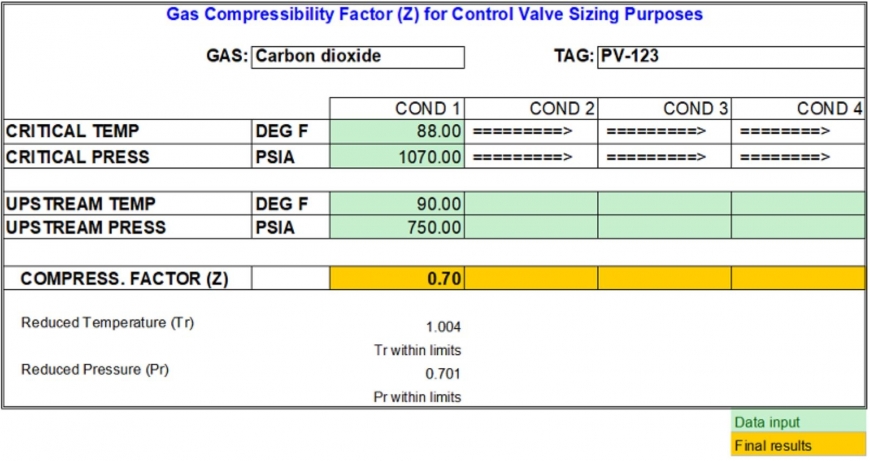
Gas Compressibility Factor and Control Valve Sizing
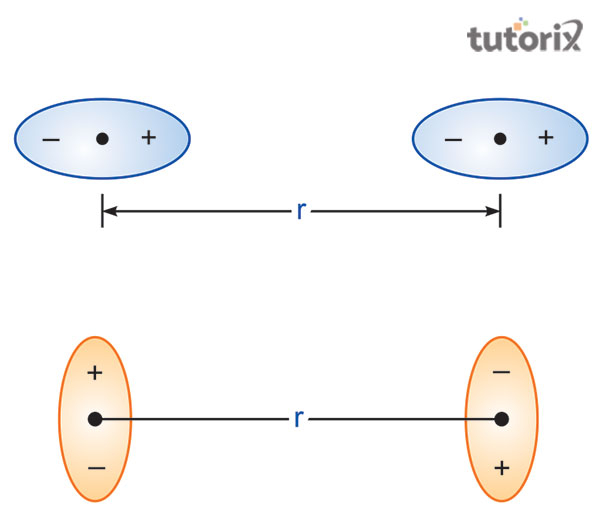
Derivation of Van Der Waals Equation

SOLVED: If PR=3, TR=2.0, the compressibility factor (Z) is equal

SOLUTION: Dpp 7 gaseous state and chemical energetics - Studypool
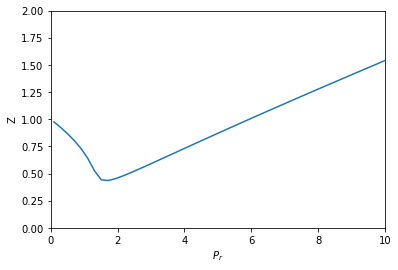
Compressibility factor variation from the van der Waals equation

Van der Waals equation - Wikipedia

Equation of state (excess compressibility factor Z À1 ¼ PV/(NkT) À

If `Z` is a compressibility factor, van der Waals' equation at low