OneClass: For a real gas, the compressibility factor, Z, is
$ 26.00 · 4.9 (688) · In stock

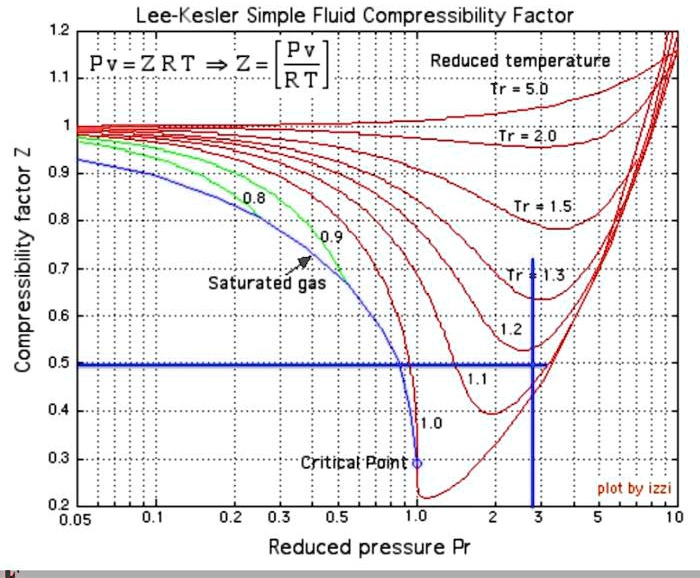
SOLVED: Lee-Kesler Simple Fluid Compressibility Factor 1.2: Pv RT Pv=ZRTZ 1.1 Reduced temperature 5.0 = 2.0 N 0.9 0.8 Compressibility factor 0.7 0.6 0.5 Tr = 0.8 0.9 Saturated gas 3 . N

Real gasses For an ideal gas, the compressibility factor Z = PV/nRT is equal to unity for all conditions. For a real gas, Z can be expressed as a function. - ppt

For an ideal gas, the value of compressibility factor `Z(=(pVm)/(RT))` is

Compressibility factor - Wikipedia

Compressibility factor - Wikipedia

Compressibility Factor Calculator

Non-Ideal Gas Behavior Chemistry: Atoms First

Compressibility factor - Wikipedia

3.2 Real gas and compressibility factor – Introduction to Engineering Thermodynamics