Slow, tight-binding inhibition of PDF by actinonin induces
$ 27.99 · 5 (313) · In stock


A [(32)P]NAD(+)-based method to identify and quantitate long residence time enoyl-acyl carrier protein reductase inhibitors. - Abstract - Europe PMC

Frédéric Dardel's research works Paris Descartes University, Paris and other places

Accurate quantitative determination of affinity and binding kinetics for tight binding inhibition of xanthine oxidase - ScienceDirect

Sonia Fieulaine's research works Université Paris-Saclay, Paris and other places

Isabelle ARTAUD, Research Director, PhD, French National Centre for Scientific Research, Paris, CNRS, Chemistry and Biology Interdisciplinar Centre paris Descartes University france

Slow, tight-binding inhibition of PDF by actinonin induces

WO2006077139A2 - Peptide deformylase inhibitors, their use, and pharmaceutical compositions containing the same - Google Patents

Marine Drugs, Free Full-Text
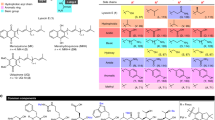
A unique peptide deformylase platform to rationally design and challenge novel active compounds

Thierry MEINNEL, Director, PhD, French National Centre for Scientific Research, Paris, CNRS, Institute for Integrative Biology of the Cell

A [(32)P]NAD(+)-based method to identify and quantitate long residence time enoyl-acyl carrier protein reductase inhibitors. - Abstract - Europe PMC
JCM, Free Full-Text

Peptide Deformylase Inhibitors as Antibacterial Agents: Identification of VRC3375, a Proline-3-Alkylsuccinyl Hydroxamate Derivative, by Using an Integrated Combinatorial and Medicinal Chemistry Approach

Carmela GIGLIONE, Laboratory Head, PhD, Université Paris-Saclay, Paris

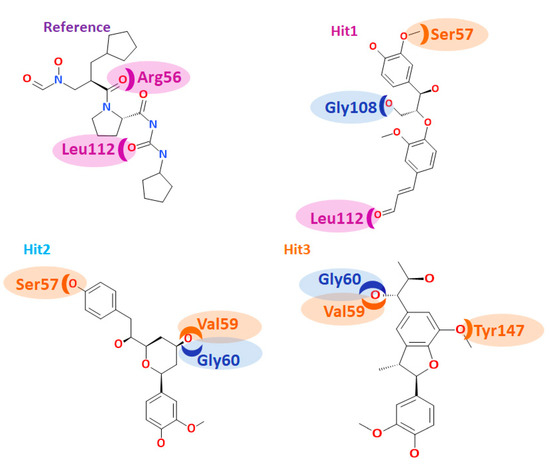
Binding mode of various PDFIs, with various scaffolds.: Binding mode of