Microbiological Media Management - SOP & Guideline - Pharma Beginners
$ 12.00 · 4.5 (487) · In stock

Standard Operating Procedure (SOP) and Guideline for the Receipt, Storage, Preparation, Growth Promotion Test, use, and Disposal of microbiological media.
SOP On Procedure For Microbiological Monitoring of Purified Water in Pharmaceutical Company, PDF, Quality Assurance

A Guide On What SOPs are Required for a New Business
SOP For Microbiological Good Laboratory Practices
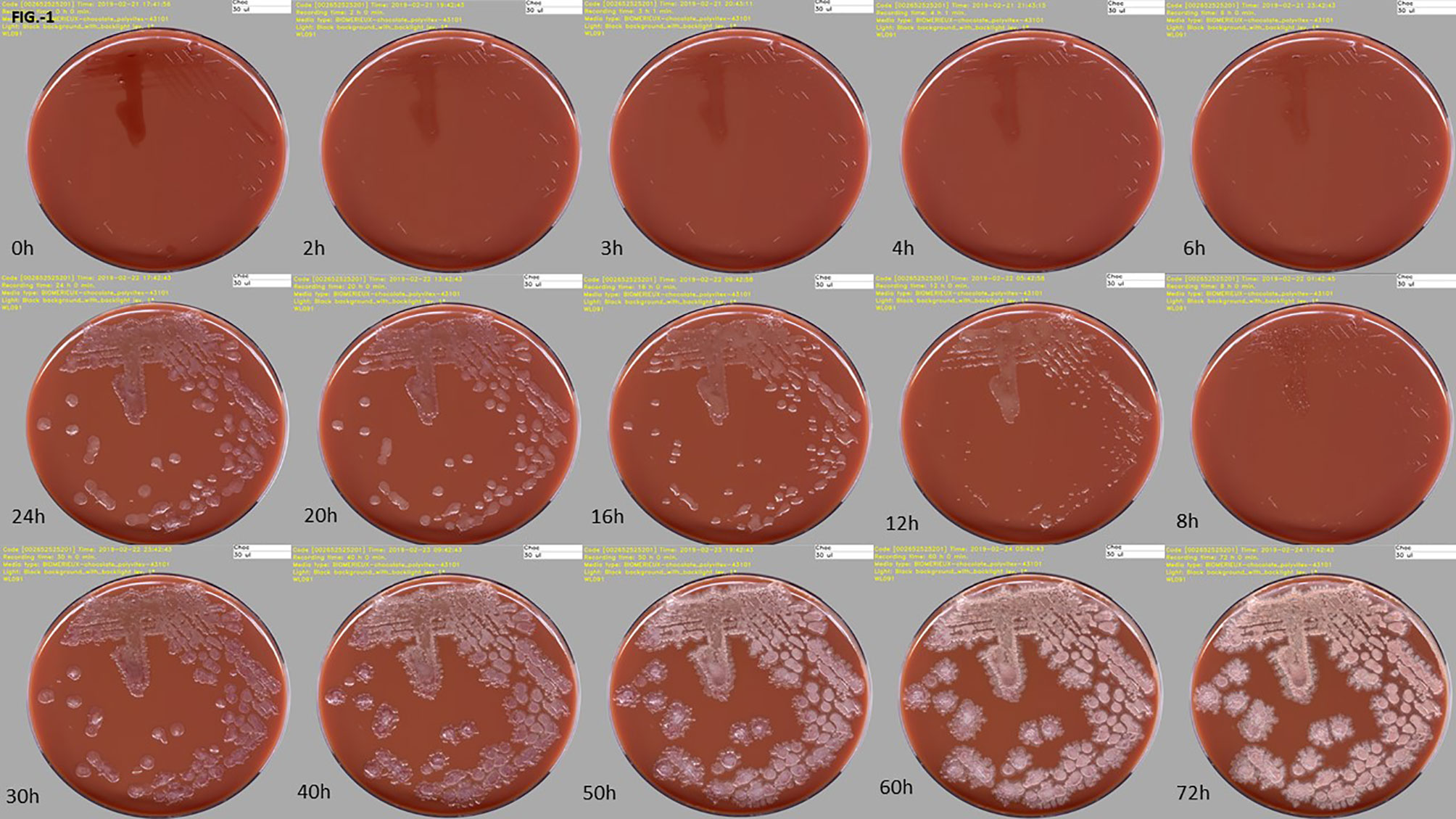
Frontiers Total Laboratory Automation for Rapid Detection and Identification of Microorganisms and Their Antimicrobial Resistance Profiles

MASTERCLAVE® Media Preparator

PDF) Preparing SOP for Microbiology Laboratory: A Short Guideline

In-process Microbial Control During Aseptic Processing (High-Risk

What is GMP, Good Manufacturing Practices
Entry and Exit of Micro Lab Sop, PDF, Clothing
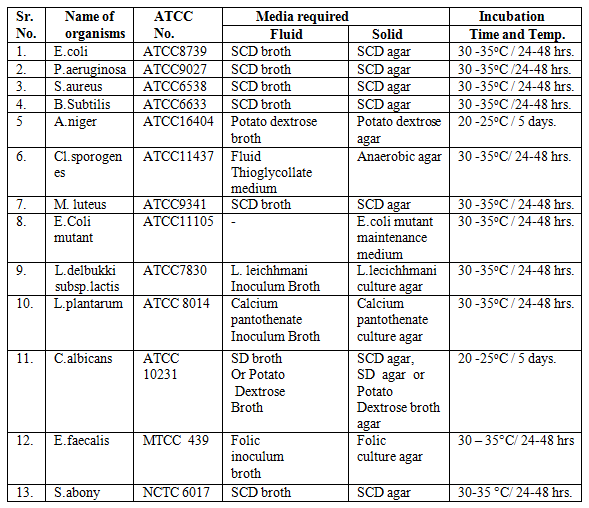
SOP for Maintenance and Transfer of Stock Cultures : Pharmaguideline

Guidelines for Temperature Control of Drug Products during Storage and Transportation (GUI-0069)

Good Laboratory Practices (GLP) - SOP & Guideline - Pharma Beginners


