FDA Approves Senza®, Nevro's High Frequency Spinal Cord
$ 6.99 · 4.8 (118) · In stock
The Senza System has been approved by the FDA for the treatment of chronic pain associated with painful diabetic neuropathy.
Nevro Corp. - AU - Relief, Multiplied

Nevro (NVRO): FDA Approved Lower Back Pain Relief Without Opiates Or Surgery, A Hold
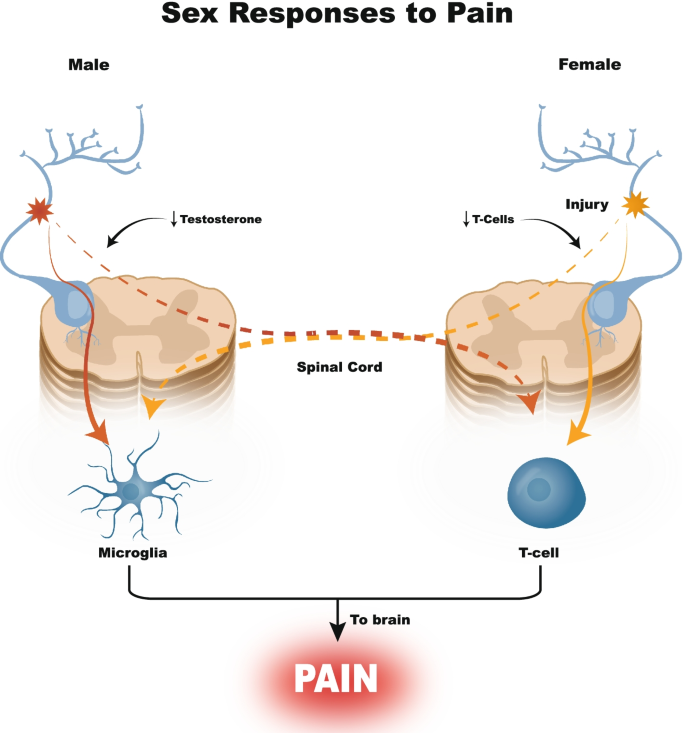
Sex-specific differences in the efficacy of traditional low frequency versus high frequency spinal cord stimulation for chronic pain, Bioelectronic Medicine

Nevro Corp Receives FDA Approval For Senza System
Endoscopy Archives - NS Medical Devices
FDA Approves Senza, Nevro's High Frequency Spinal Cord Stimulation Therapy For PDN - Beyond Type 1
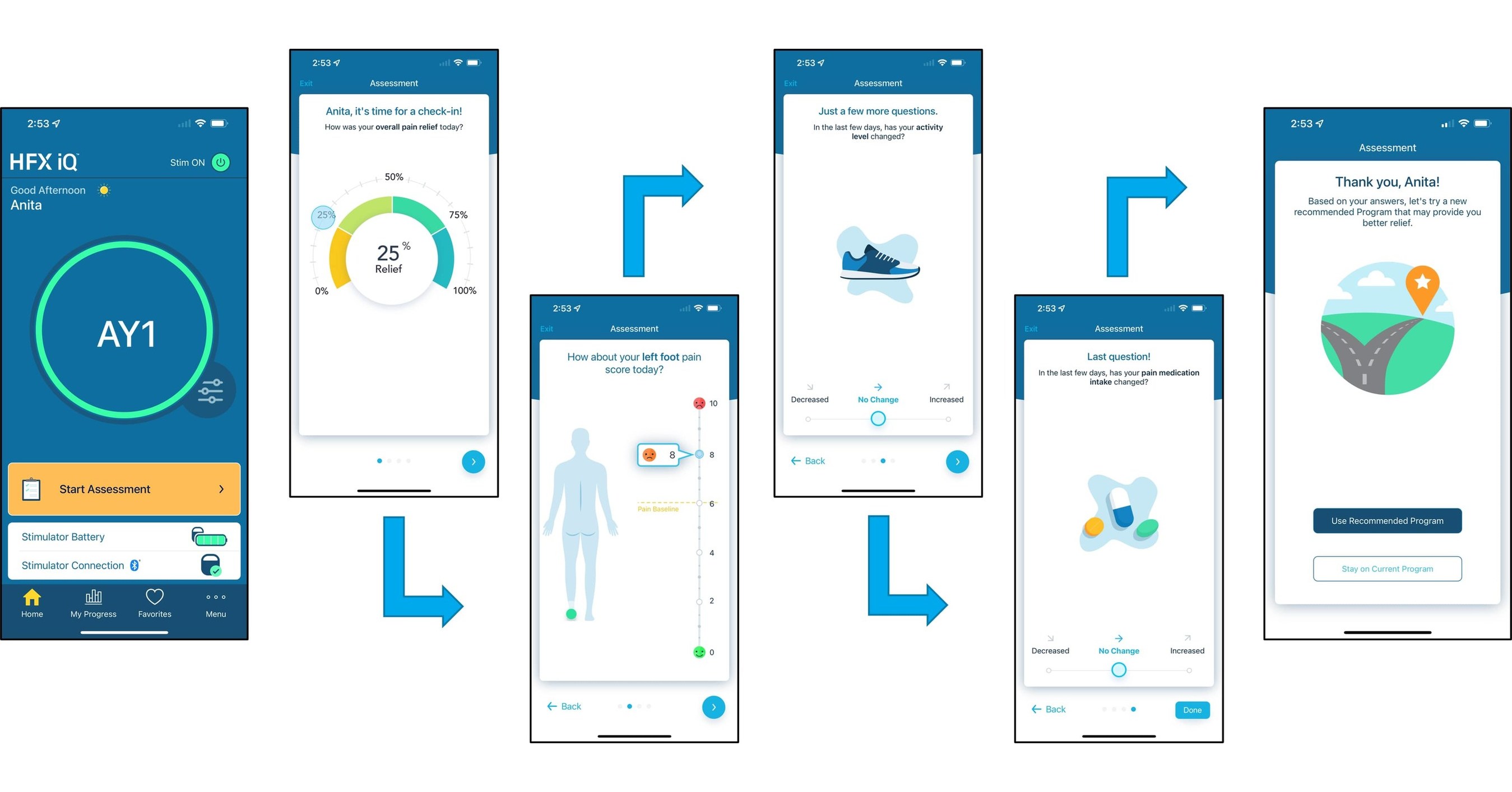
Nevro Announces FDA Approval of HFX iQ™ Spinal Cord Stimulation System to Personalize the Treatment of Chronic Pain
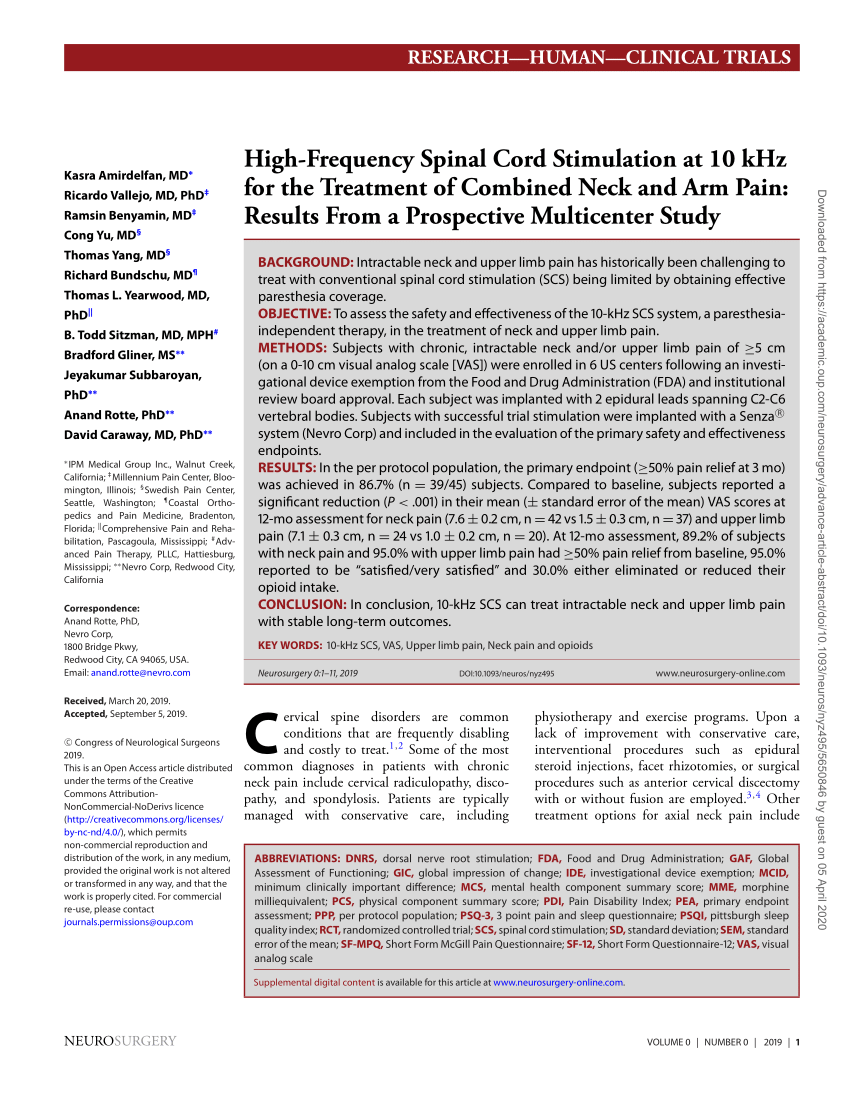
PDF) High-Frequency Spinal Cord Stimulation at 10 kHz for the Treatment of Combined Neck and Arm Pain: Results From a Prospective Multicenter Study
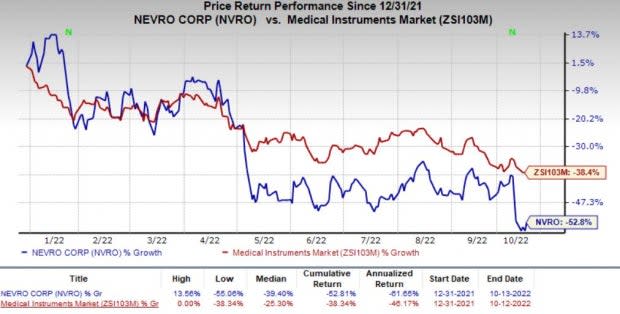
Nevro (NVRO) Gains Following FDA Approval for Senza HFX iQ
HF10 Therapy Shown Effective For Variety of Pain Conditions

Nevro Spinal Cord Stimulation in Bellevue and Seattle
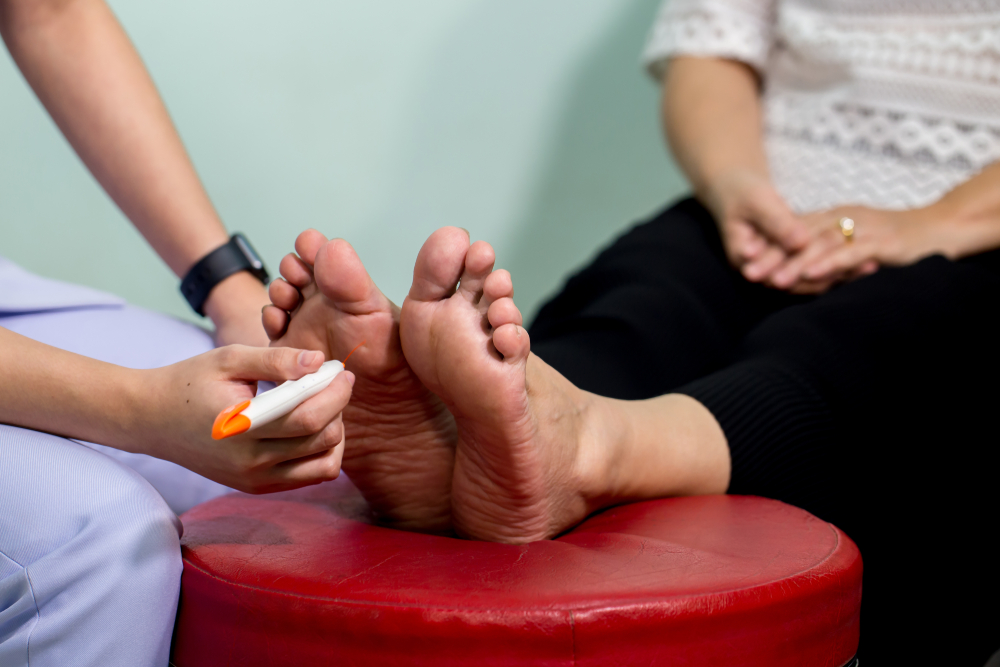
Nevro Study on Painful Diabetic Neuropathy and Spinal Cord Stimulation - Colorado Pain Care

Nevro announces FDA approval of Senza SCS system for PDN
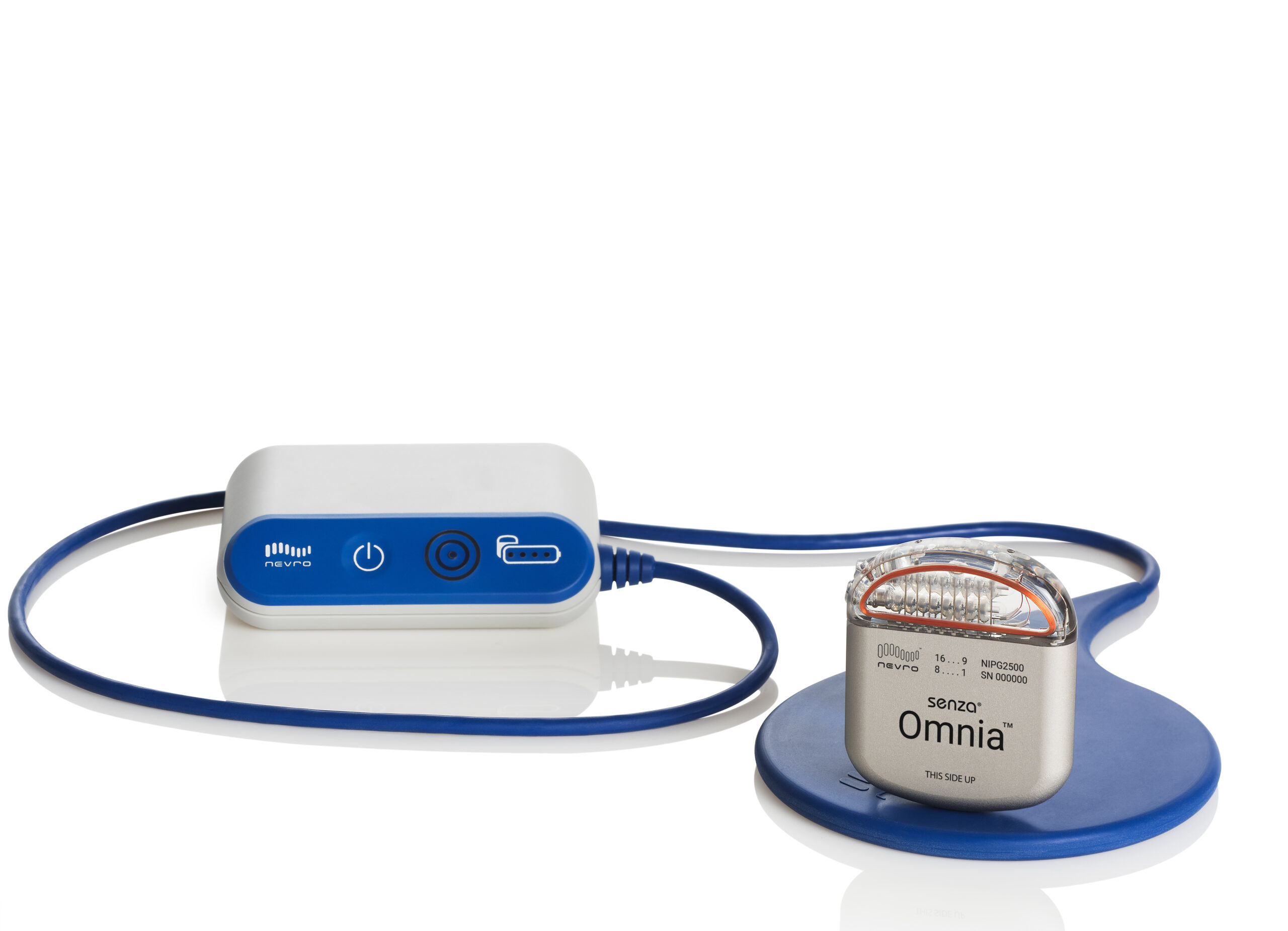
Which Spinal Cord Stimulator is Best?