Applications for Medical Device Investigational Testing Authorizations Guidance Document
$ 15.99 · 4.8 (162) · In stock

Applications for Medical Device Investigational Testing Authorizations Guidance Document
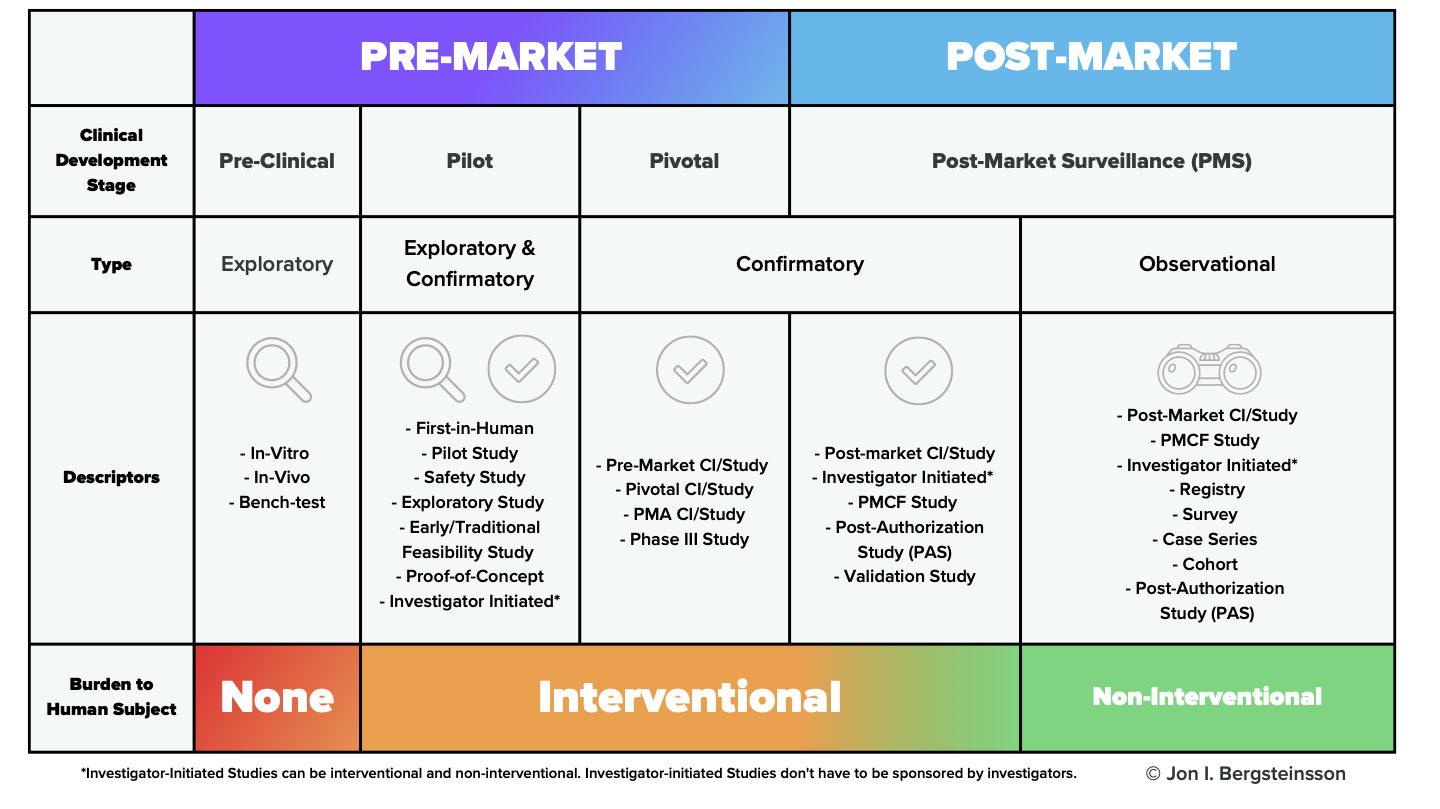
ISO 14155:2020 Compliant Clinical Investigations - The Ultimate Guide

Analysis of U.S. Food and Drug Administration Draft Guidance on Clinical Trials with Psychedelic Drugs
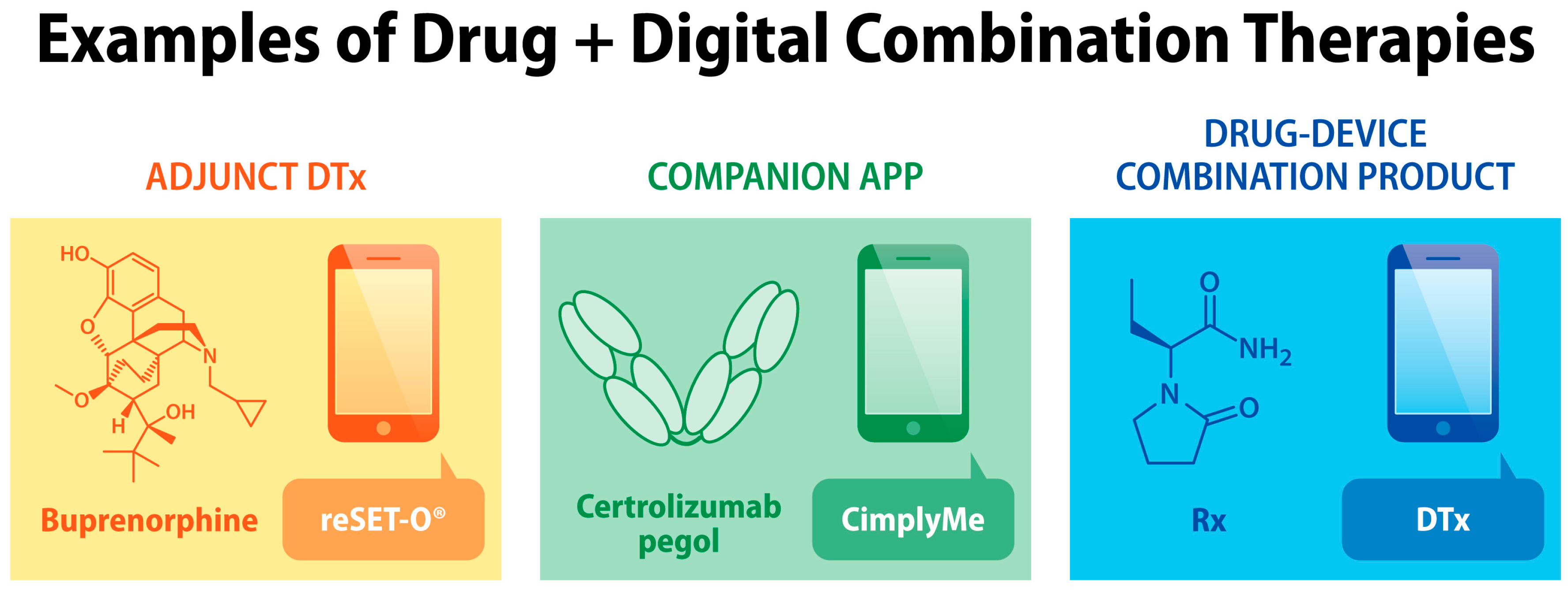
JCM, Free Full-Text

Medical Device Regulations and Guidelines

Health Canada Creates the Medical Device Directorate

Health Canada Approval Process for Medical Devices: Step-by-Step Guide

FDA Seeks Input on Intended 2016 Guidance Documents
List of documents to upload to the Spanish Agency of Medicines and
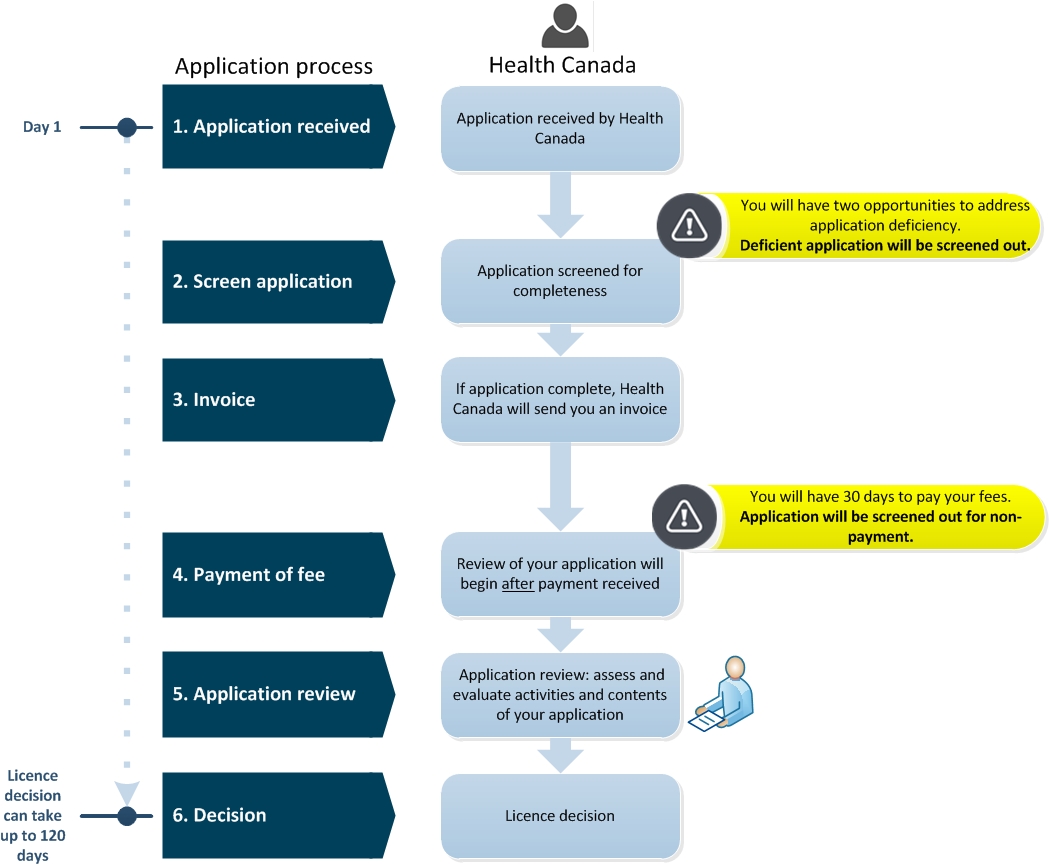
Guidance on Medical Device Establishment Licensing (GUI-0016

Class I and Class II medical devices: defining, classifying and marketing them