FDA Issues New Mammography Guidelines for Women With Dense Breasts
$ 14.99 · 4.8 (189) · In stock
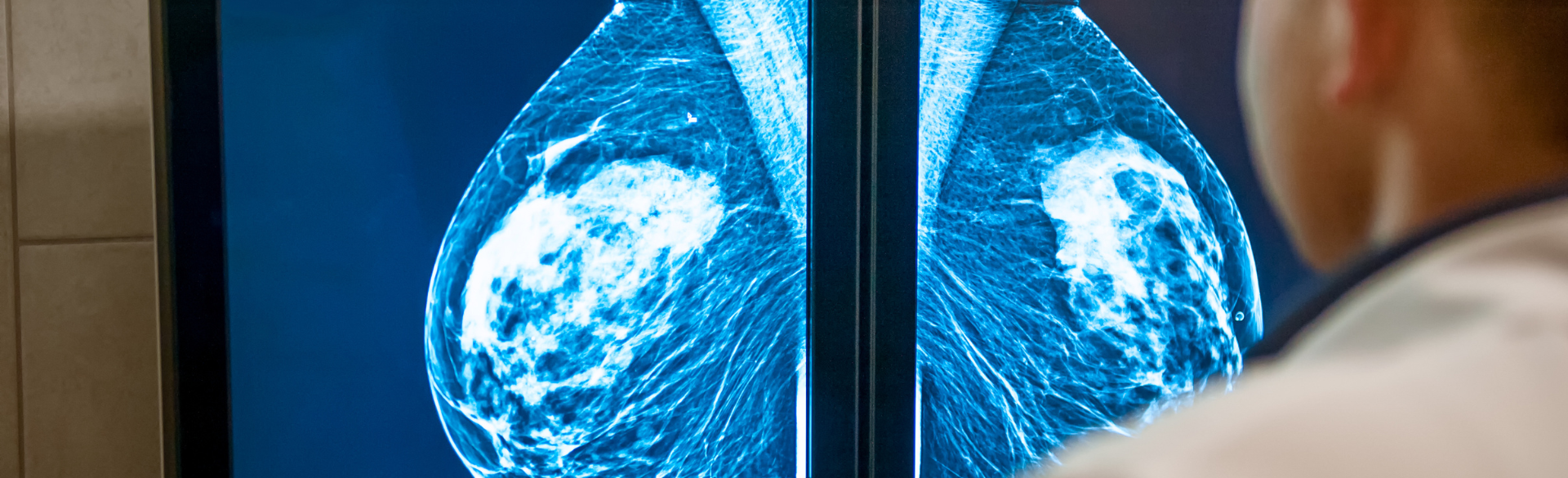
The FDA on March 9 updated its mammography guidelines to require mammography facilities to notify patients about the density of their breasts.
New breast cancer screening guidelines call for women to start mammograms at age 40

Why the FDA is tightening mammogram requirements — Harvard Gazette

FDA Announces New Mammogram Standards for People With Dense Breasts

FDA Will Require Dense Breast Disclosure at Mammogram Clinics - The New York Times
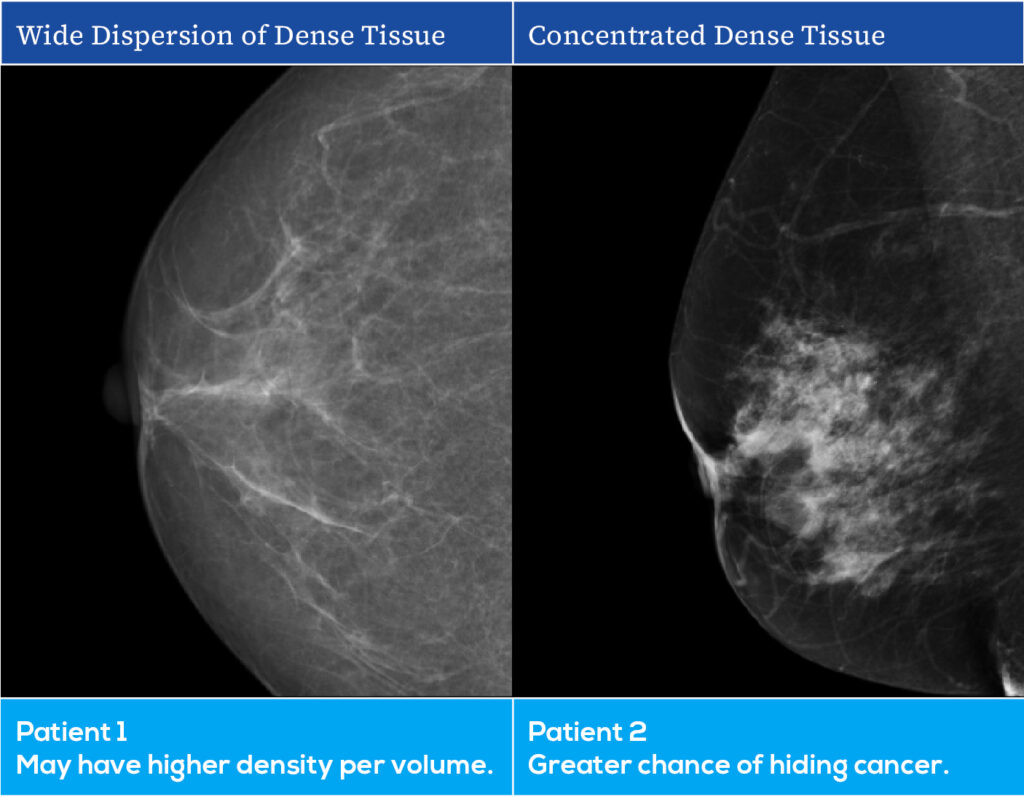
AI Breast Density Assessment
![]()
CU Cancer Center
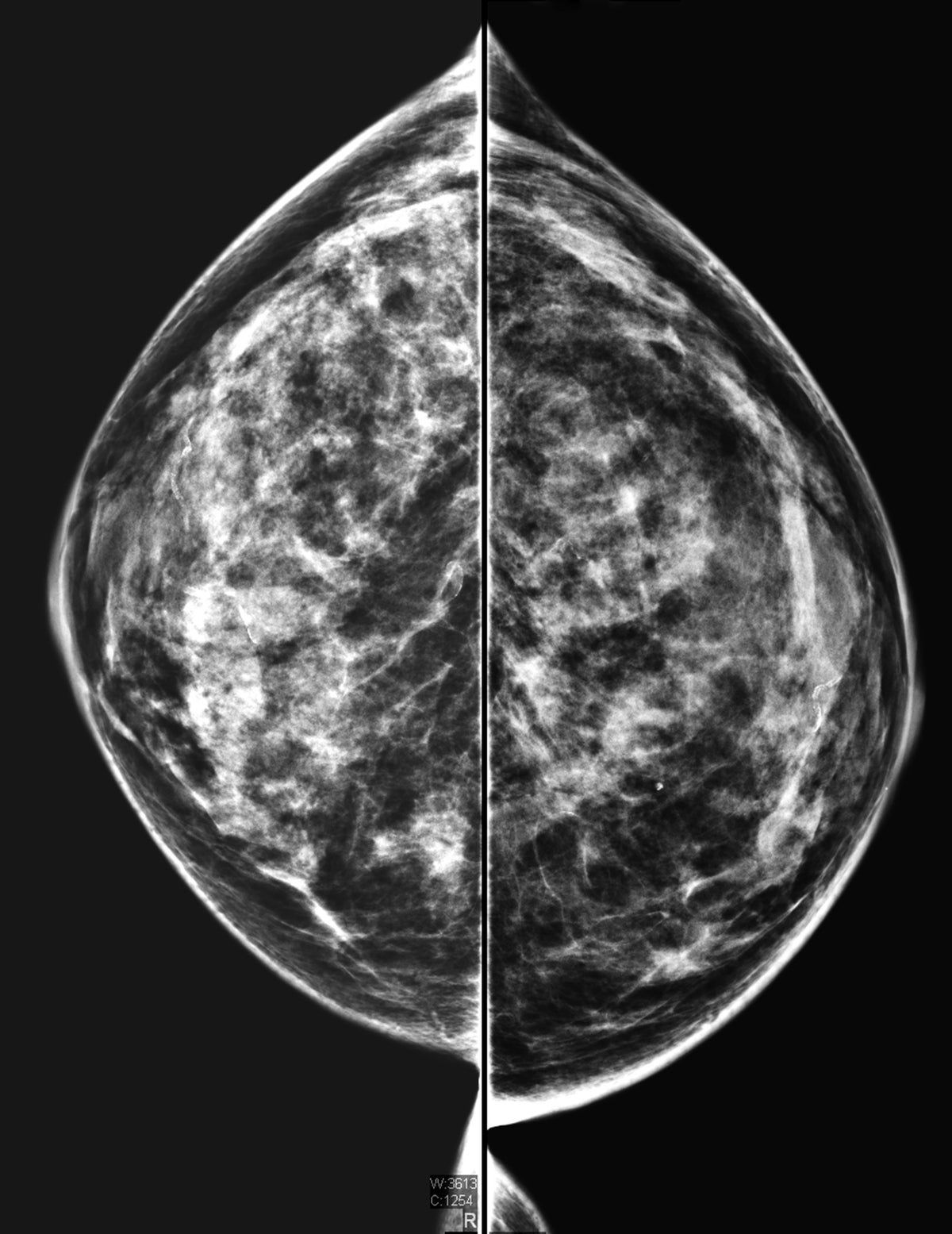
What the FDA Ruling about 'Dense Breasts' Means for Cancer Risk and Screening

Medical practitioners will have to notify patients about breast density in mammograms under new FDA regulations - CBS News
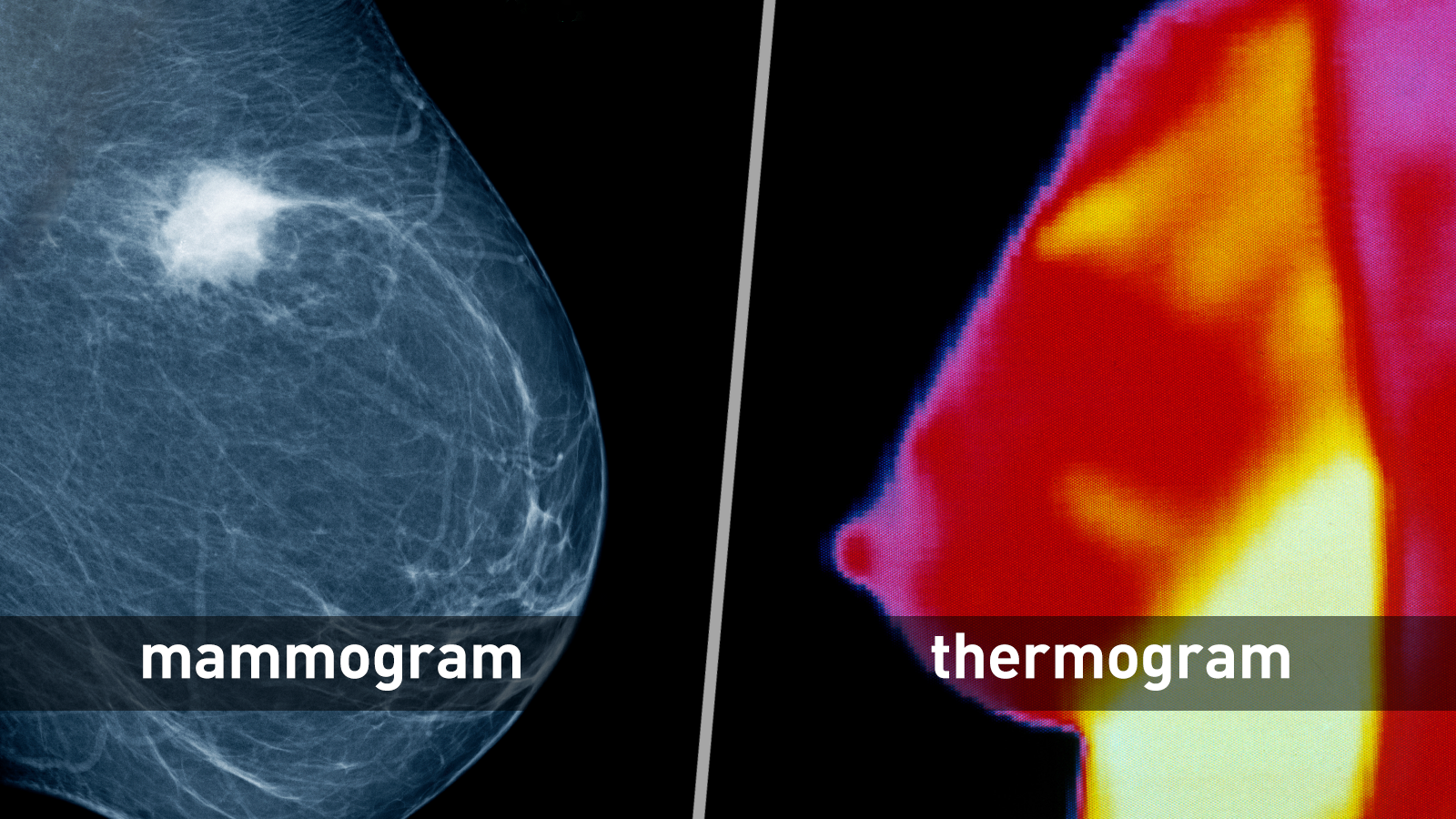
Breast Cancer Screening: Thermogram No Substitute for Mammogram

FDA Issues New Mammography Guidelines for Women With Dense Breasts

CU Cancer Center

CU Cancer Center
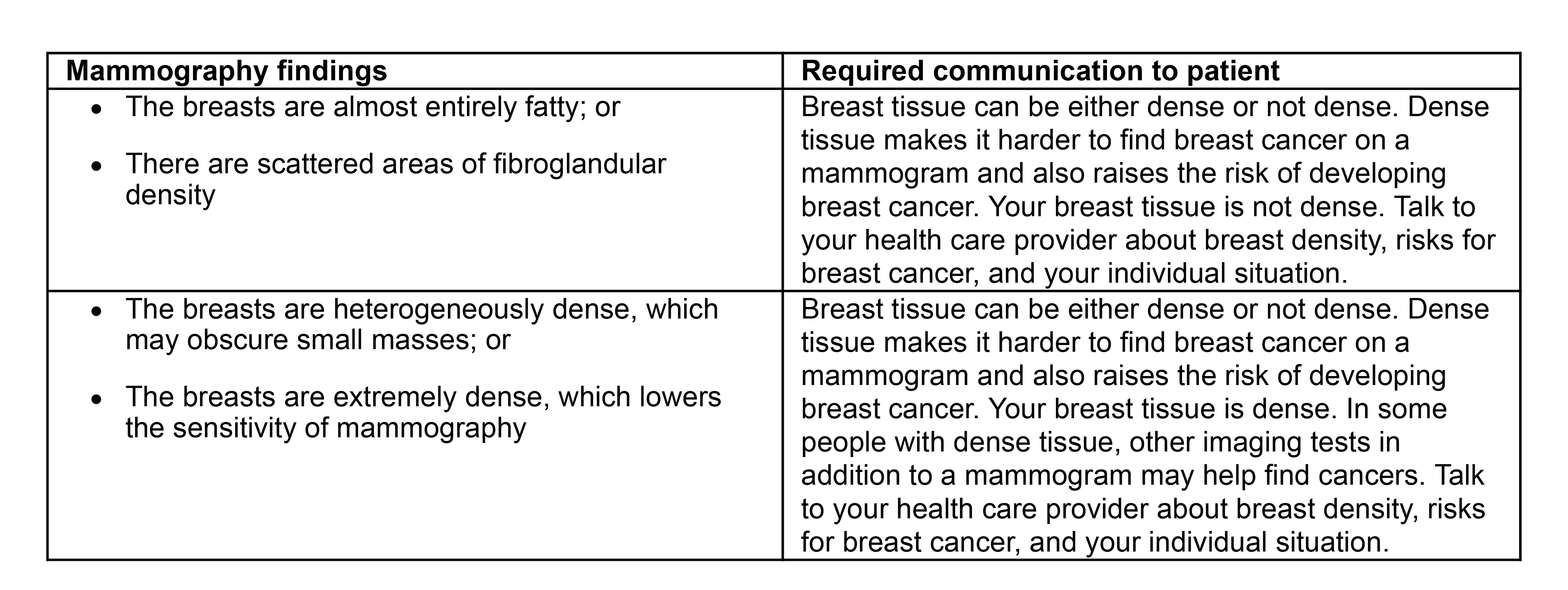
Major updates coming to mammography quality and certification standards including new dense breast notification requirements - Lexology
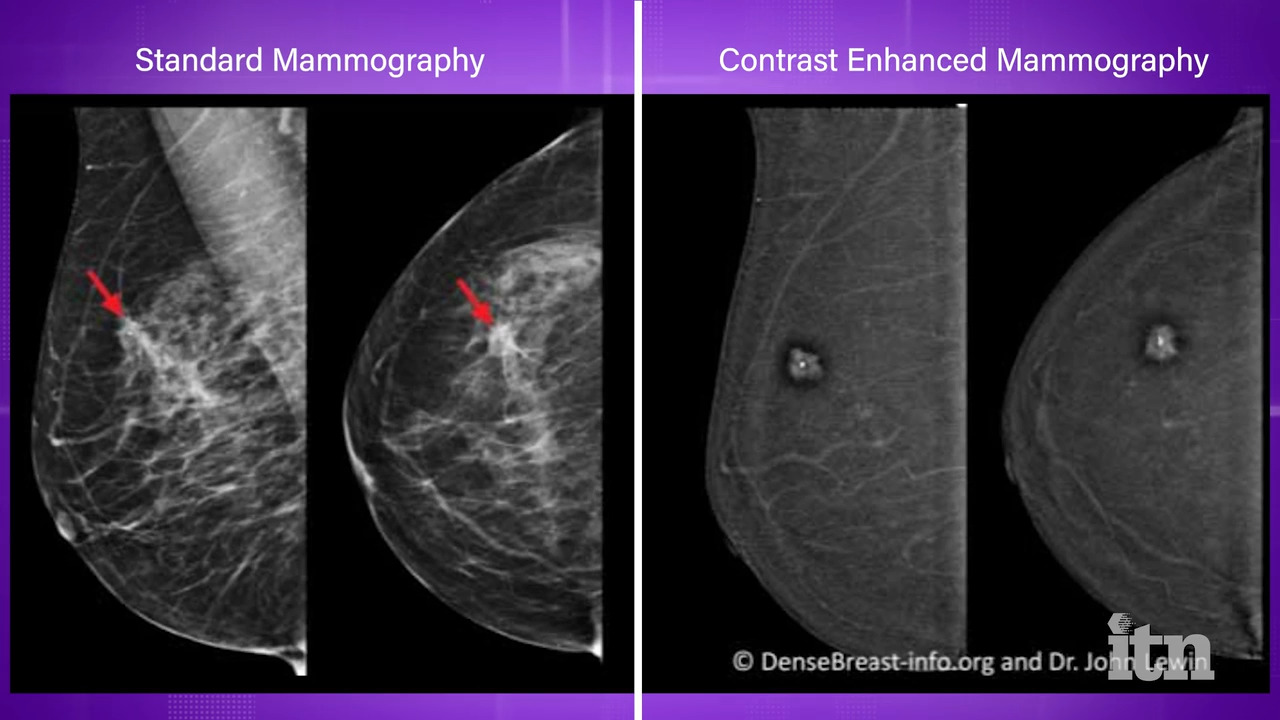
Videos Imaging Technology News

Diagnostic Performance of Digital Breast Tomosynthesis for Breast Suspicious Calcifications From Various Populations: A Comparison With Full-field Digital Mammography - ScienceDirect