Indian industry group calls for clearer expiry date labelling rules for topic drugs
$ 10.50 · 4.8 (572) · In stock

The DCGI needs to clarify expiry date labelling rules for topical drugs according to an Indian trade group, which says some members’ products are being deemed to be ‘misbranded.’

labelling means a display of written, printed or graphic matter upon immediate container or the wrapper of a drug package o

Indian industry group calls for clearer expiry date labelling rules for topic drugs

US FDA's Hamburg offers few details on trip to India

Nutrition facts label - Wikipedia

India could see drug shortages, price rise owing to new rule: Industry executives - The Economic Times

Orsl: Devil lies in the details: ORSL sneaks into chemist shelves in a new avatar

2024 FDA Cosmetic Labeling and Packaging Requirements

French regulator raises concerns over Indian CRO studies
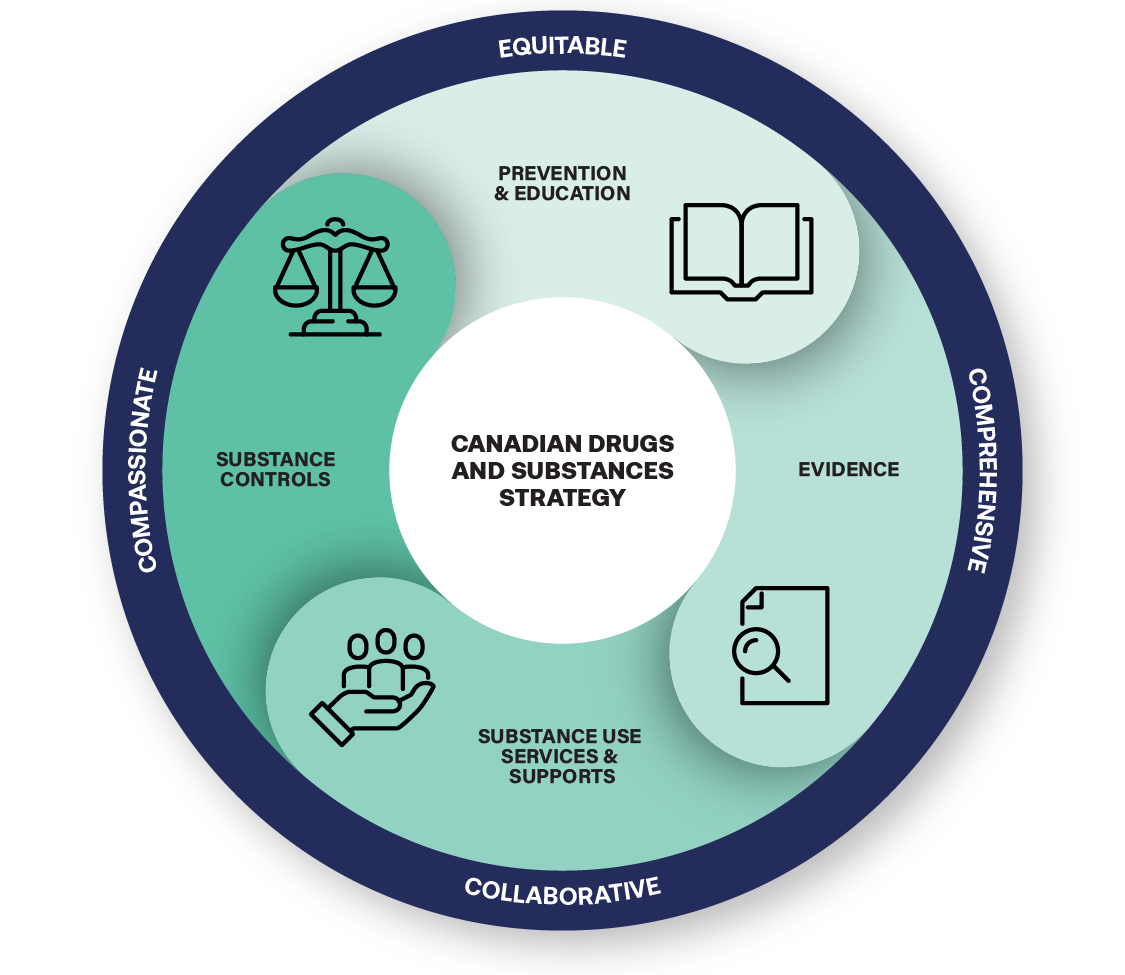
Canadian Drugs and Substances Strategy: Overview

All India Active Pharma Ingredient & Bulk Drug Directory 2023 - 24 by The Mazada Pharma Guide - Issuu