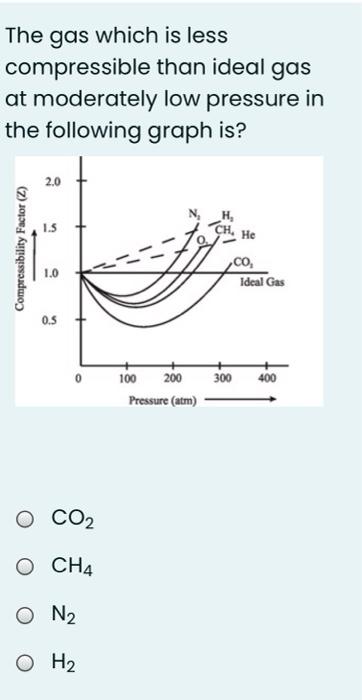
In the following compressibility factor Z vs pressure graph at 300 K, the compressibility of CH 4 at pressure
$ 13.99 · 4.8 (292) · In stock

In the following compressibility factor Z vs pressure graph at 300 K, the compressibility of CH 4 at pressure
In the following compressibility factor Z vs pressure graph at 300 K- the compressibility of CH 4 at pressure -200 bar deviates from ideal behaviourA- The molar volume of CH 4 is less than its molar volume in the ideal stateB- The molar volume of CH 4 is same as that in its ideal stateC- Intermolecular interactions between CH 4 molecules decresasesD- The molar volume of CH 4 is more than its molar volume in the ideal state

A real gas M behaves almost like an ideal gas. Graph 1 is obtained by plotting volume, V against temperature, T for x mol of gas M at pressure, P_1. a. Suggest
The volume vs. temperature graph of 1 mole of an ideal gas is
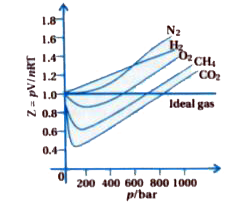
Gujrati] Explain compressibility factor (Z).
Is z (compressibility factor) vs P (pressure) graph drawn by

Compressibility factor - Wikipedia

Energies, Free Full-Text
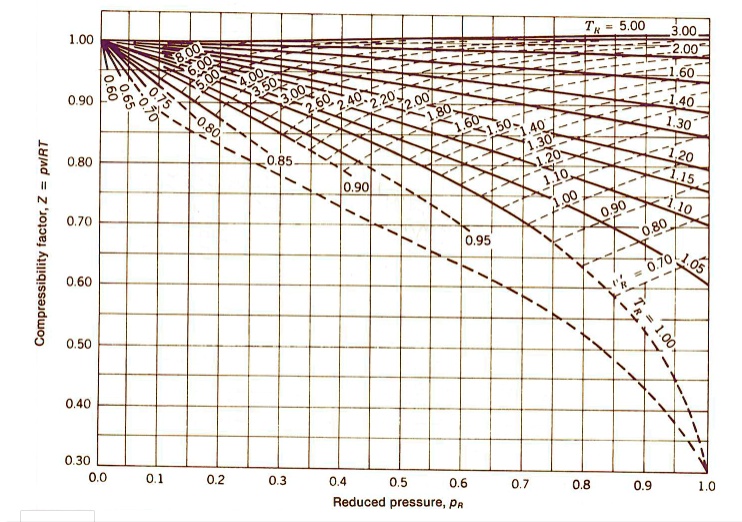
SOLVED: Determine the specific volume (m^3/kg) for air at T

Real-gas z-factor, as attributed to Standing and Katz, 9 plotted as a
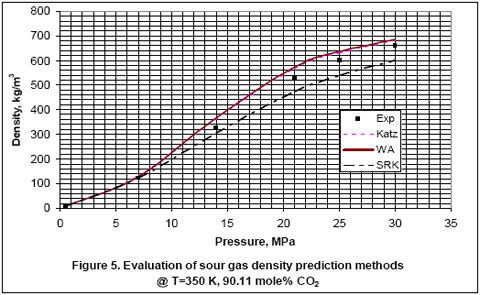
How good are the shortcut methods for sour gas density


KVPY-SX 2016 Chemistry Question Paper with Solutions PDF Download

The role of the compressibility factor Z in describing the volumetric behavior of gases
Compressibility Factor : The ratio of the observed volume of a gas
Solved The first order rate constant of the decomposition of

Compressibility factor - Wikipedia