Ideal–Universal Gas Law
$ 20.50 · 4.7 (317) · In stock

Definition: The Universal or Ideal Gas Law describes the relationship between all four properties (pressure, volume, number of moles, and temperature) as well as a gas constant called “R.” NOTE: The Ideal Gas Constant “R” has constant a value of 0.0821 L.atm/mol.K Relation: The relation between pressure (P) volume (V), number of moles (n) and…
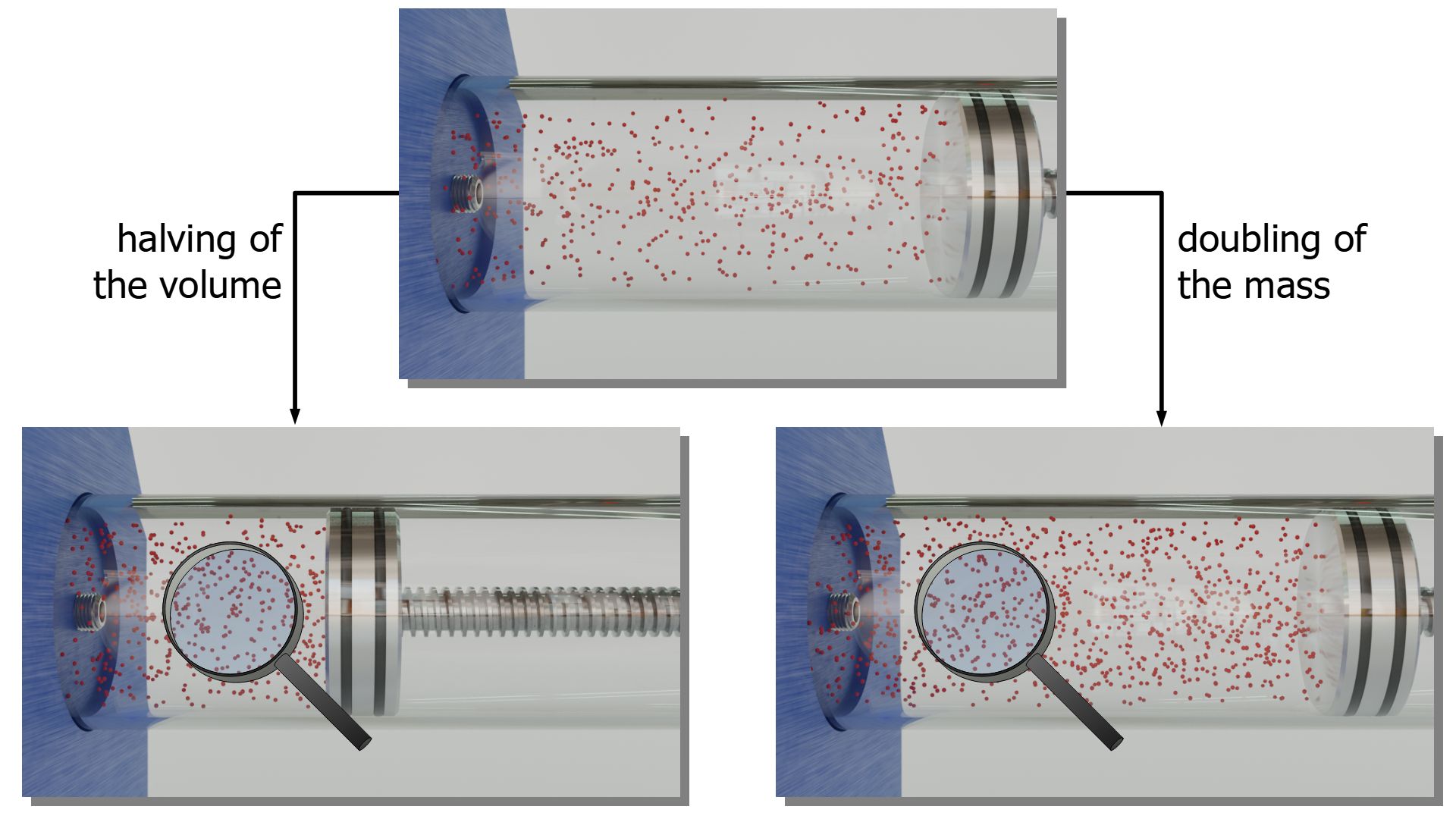
Ideal gas law (explained and derived) - tec-science

Forming Ions for Bonding

Intermolecular Forces of Attraction
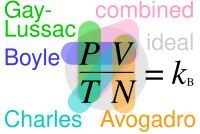
Ideal gas law - Wikipedia

Combined Gas Law, Definition, Formula & Example - Lesson

Combined Gas Law

Forming Compounds

Equation Of State (Ideal Gas), Glenn Research Center

Gas Constant: Definition, Formula, Ideal Gas and Examples
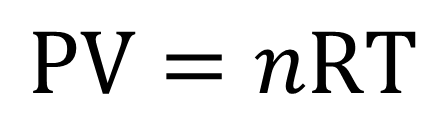
Ideal Gas Rulebreakers - HVAC School

Boyles' Law

Charles' Law

Ionization Energy

Ideal Gas Law — Overview & Calculations - Expii