Does atomic size increase down a group?
$ 9.00 · 4.5 (577) · In stock

Does atomic size increase down a group?
Does atomic size increase down a group

Why does the atomic size increases down a group? - Quora

Atomic Radius - Definition, Types, Periodic Trends of Atomic Radii, Video, and FAQs

What is a periodic property? How do the following properties change in a group and period? Explain.(a) Atomic radius (b) Ionization energy (c) Electron affinity (d) Electronegativity.

Atomic and ionic radii (video)
Atomic Structure Questions Relative Atomic Mass, Relative
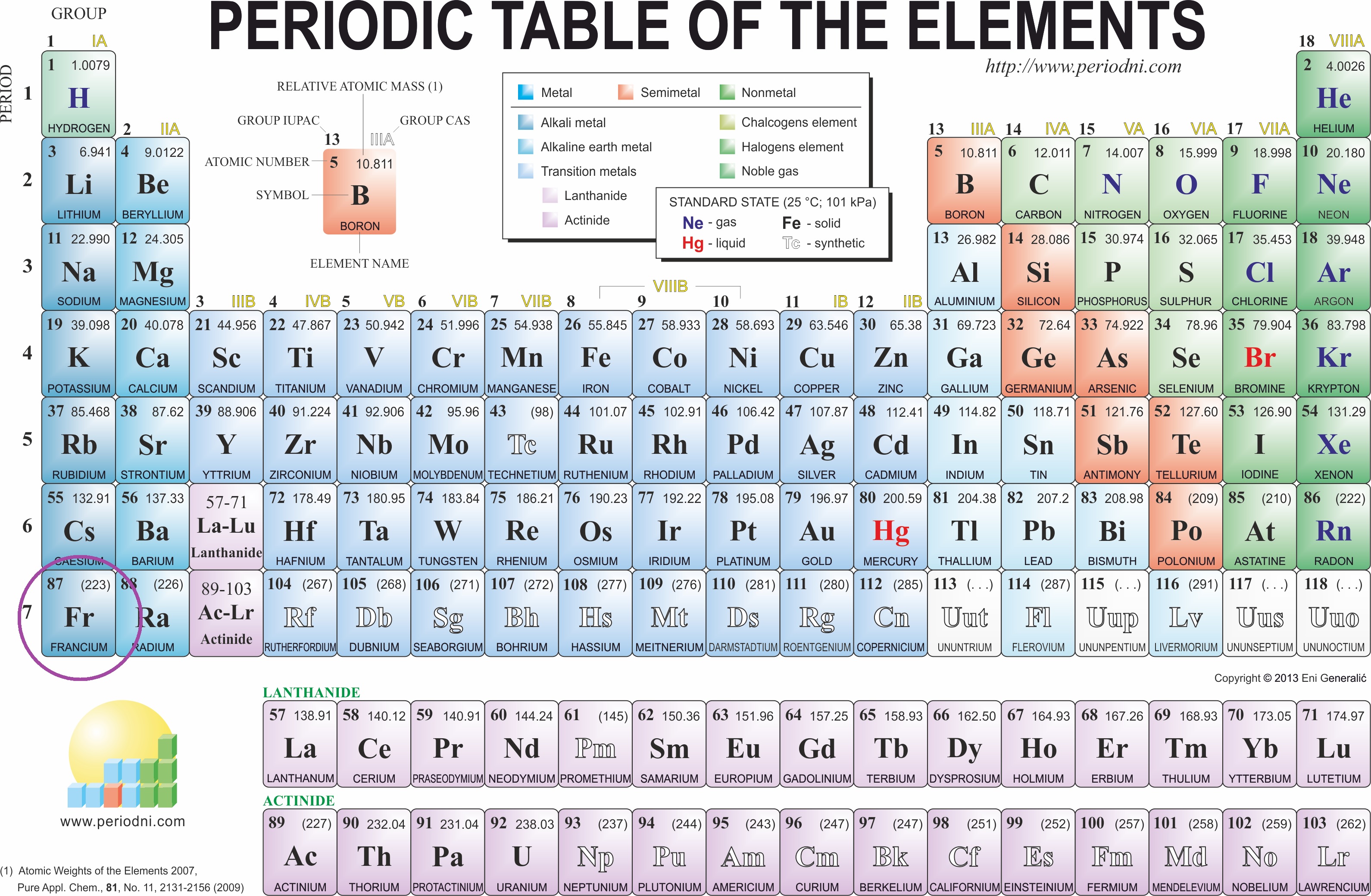
What are the group trends for atomic radius?

Periodic Properties and Trends Atomic Radii Size Increases going down a group.Size Increases going down a group. Because electrons are added further. - ppt download
Why does ionic radii increase down the group in the periodic table
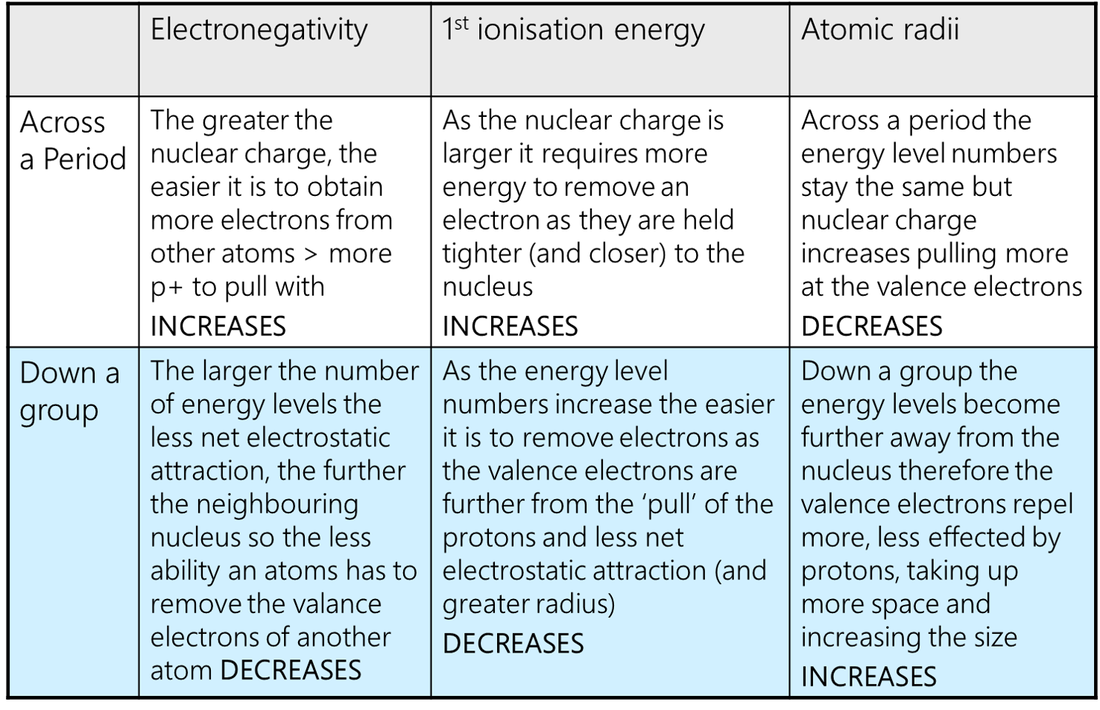
2. Periodic Trends

SOLUTION: Atomic sizes, Atomic radii, covalent radii, ionic radii
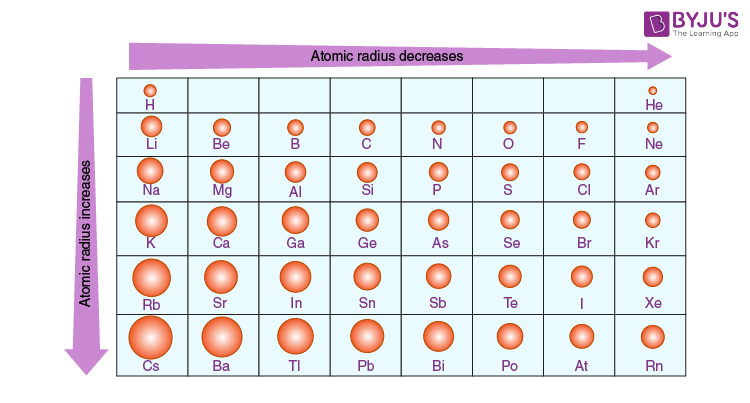
Atomic Size (Atomic Radius) - Definition & Variation in Periodic Table with Videos of Atomic Radius

Physics Revision Notes, Physics - STPM

Periodicity 3: Why Atomic Radii Increases Down a Group
