Compressibility factor Z - Gaseous State
$ 10.00 · 4.6 (780) · In stock

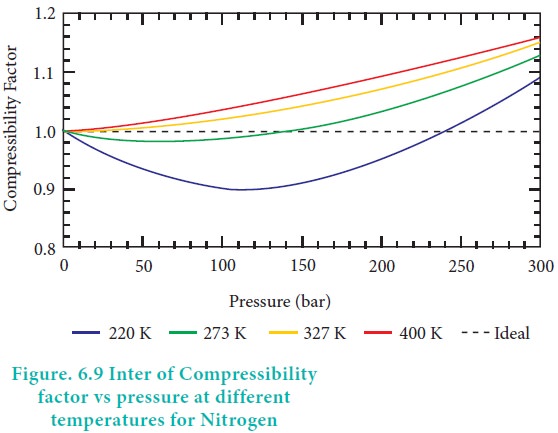
Compressibility factor Z - Gaseous State

The compressibility factor Z for the gas is given by
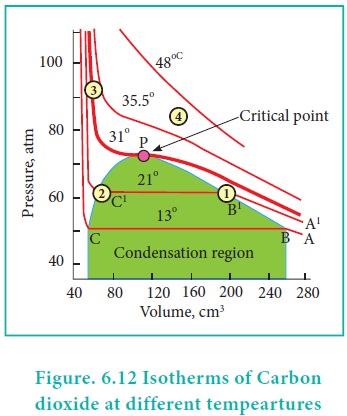
Pressure-Volume isotherms of Carbon dioxide

The Gas Laws
Ideal gases and real gases are compressible or not compressible what is the compressible factor for real gases and ideal gases.

The compressibility factor for an ideal gas is

physical chemistry - Is the compressibility factor smaller or greater than 1 at low temperature and high pressure? - Chemistry Stack Exchange
Is z (compressibility factor) vs P (pressure) graph drawn by changing volume? If it is why it isn't drawn by changing mole - Quora

1.1: Thermodynamic Variables and Equations of State - Chemistry LibreTexts

Compressibility Factor Z Important Concepts and Tips for JEE Main

Real Gas Behavior The Compression Factor (Z) [Example #2]

Compressibility factor, Z of a gas is given as Z= frac { pV }{ nRT