FDA Cleared vs Approved vs Granted for Medical Devices
$ 22.99 · 4.7 (600) · In stock
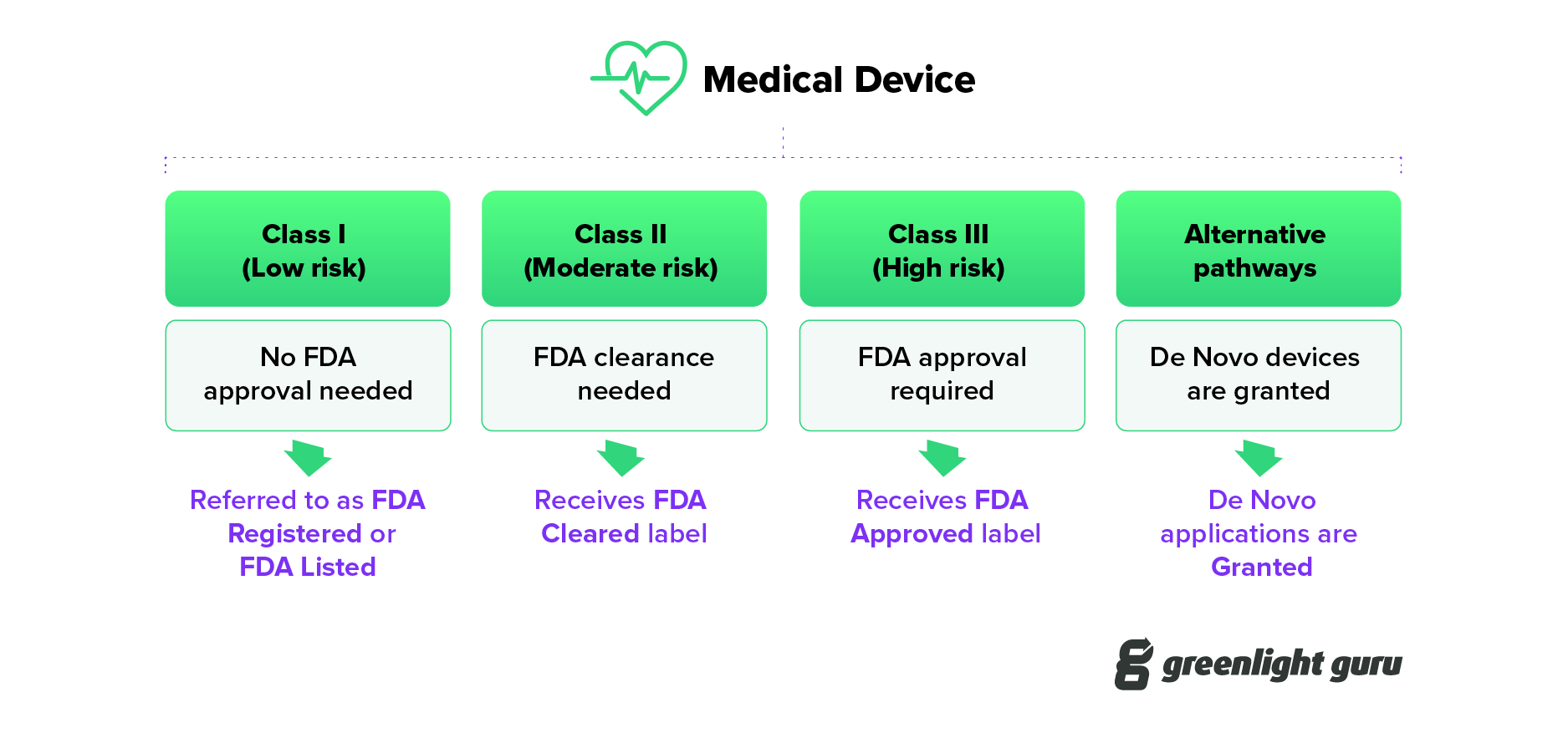
Ever wonder what FDA cleared vs approved vs granted actually mean? Learn the subtle yet important differences between these regulatory terms.

Understanding FDA Registered vs Cleared vs Approved vs Granted for Medical Devices
Your Medical Devices Are Getting Smarter. Can the FDA Keep Them Safe? - WSJ

Beyond the 510(k): The regulation of novel moderate-risk medical devices, intellectual property considerations, and innovation incentives in the FDA's De Novo pathway

For FDA Approval, It All Boils Down to the Validation - Medical Design Briefs

What it Means to be Focused on Quality vs. Compliance

The FDA submission process: 510K vs PMA. What's the difference?
FDA Listed vs. Cleared vs. Approved: What's the difference?

FDA Clearance Granted for First AI-Powered Medical Device to Detect All Three Common Skin Cancers

FDA listed, cleared, approved, granted - what IS the difference?

The Difference Between FDA Registered, Cleared, Granted, Authorized and Approved

Expedited Access Pathway

FDA Device Regulation: 510(k), PMA · Academic Entrepreneurship for Medical and Health Sciences

FDA Clearance Granted for First AI-Powered Medical Device to Detect All Three Common Skin Cancers (Melanoma, Basal Cell Carcinoma and Squamous Cell Carcinoma)
USA regulatory process for medical devices

