117. Compressibility factor H, behaving as rea gas is 1) 1 RTV 3) 1+- RT 4) (1-a) 18. If V is the observed molor unlum
$ 21.00 · 4.7 (376) · In stock

Click here:point_up_2:to get an answer to your question :writing_hand:117 compressibility factor for h behaving as reagas is1 1rtv31rt41a18 if v is the observed
Click here👆to get an answer to your question ✍️ 117- Compressibility factor H- behaving as rea gas is 1- 1 RTV 3- 1- RT 4- -1-a- 18- If V is the observed molor unlum
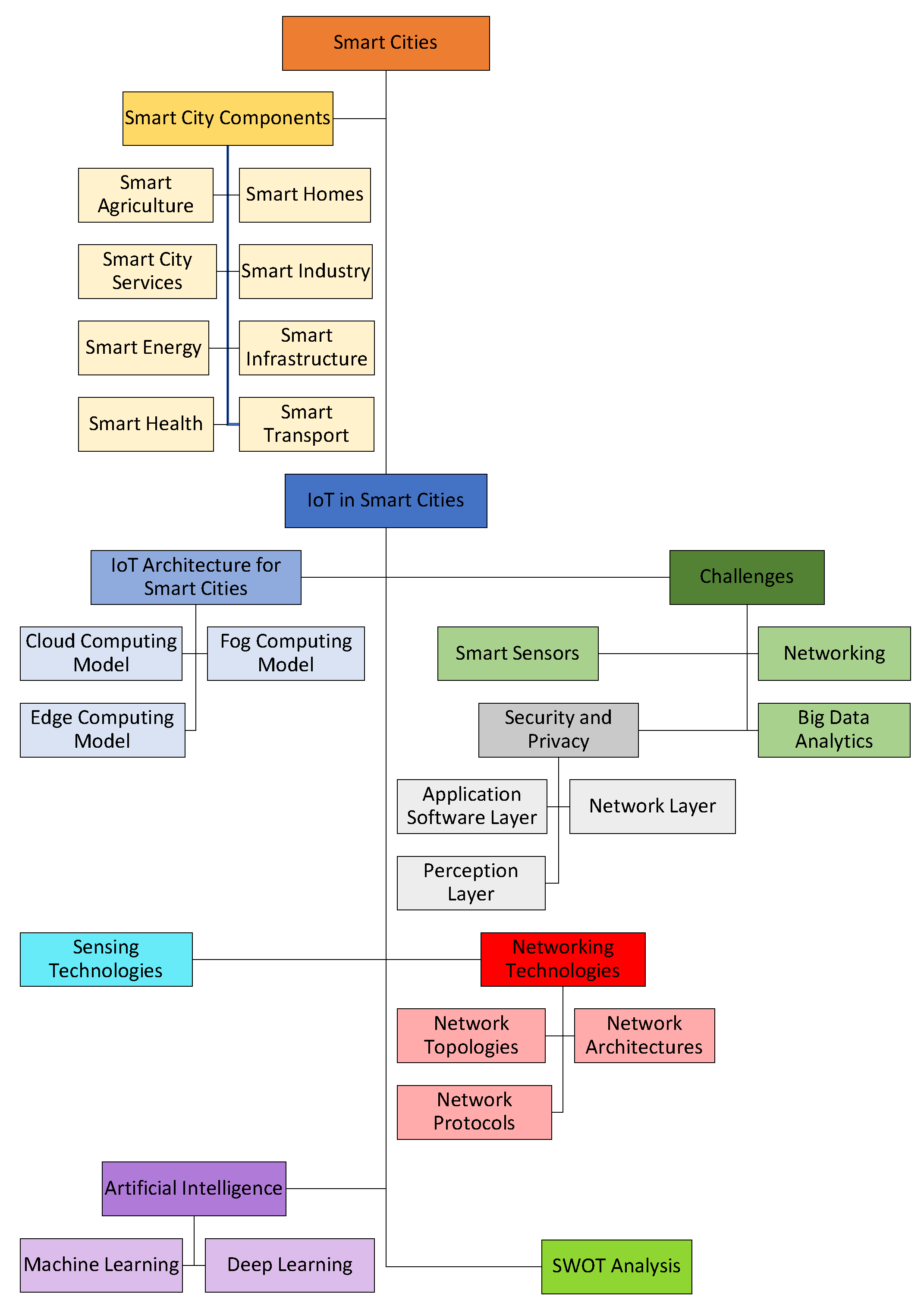
Smart Cities, Free Full-Text

014- 1-pb/RT The compressibility factor a real gas high pressure is : 1+pb/RT 1+RT/Pb
Solved The plot below shows how compressibility factor (Z)

ME2036- ENGINEERING THERMODYNAMICS BY Mr.P.SATHISH

Compressibility factor (gases)/Citable Version - Citizendium

quot; Who controls the vocabulary , controls the knowledge " - FBC>s

Determine Compressibility of Gases
Gasdynamics PDF, PDF, Mach Number
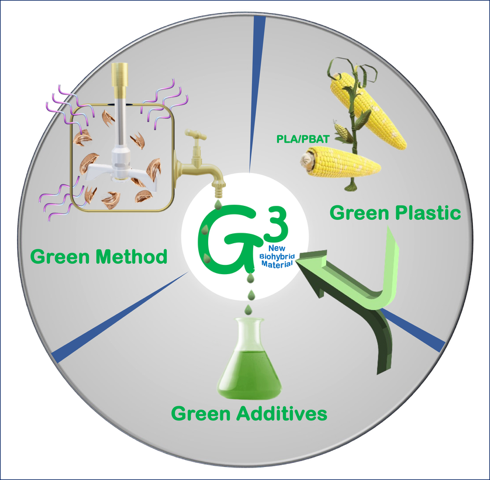
IPCB Publications Database
Physics Textbook, PDF, Torque

Solved We begin by showing that the compressibility factor

Compressibility factor for H2 behaving as real gas is

a) A gas at 250 K and 15 atm has a molar volume 12 per cent
The compression factor (compressibility factor) for one mole of a van der Waals' gas - Sarthaks eConnect
Physics Textbook, PDF, Torque



